Iscriviti per sbloccare più funzioni
- Salva questo mazzo nel tuo account
- Studia le flashcard con la ripetizione dilazionata
- Esporta in Anki (.apkg) o PDF
- Elabora documenti fino a 100 pagine
- Immagini estratte da PDF e documenti
- Estrazione testo migliorata da PDF e documenti
- Flashcard migliori con il nostro modello AI più avanzato
Flashcard in questo mazzo (51)
-
chemistry
-
Define an 'element'.
An element is a pure substance with specific physical/chemical properties that cannot be broken down into a simpler substance.
chemistry -
What is an 'atom'?
An atom is the smallest unit of matter that still retains the chemical properties of the element.
chemistry -
chemistry
-
What is a 'monomer' and a 'polymer'?
- Monomer: Single molecule that can polymerize
- Polymer: Substance made of many monomers joined in chains
macromolecules -
What occurs in a dehydration (condensation) reaction?
A covalent bond forms between monomers and water is released.
chemistry reactions -
chemistry reactions
-
What are the primary roles and elemental composition of carbohydrates?
Carbohydrates provide fuel and structural support and contain carbon, hydrogen, and oxygen (CHO).
carbs -
Name three monosaccharides and their carbon counts.
- Ribose: five-carbon
- Fructose: six-carbon
- Glucose: six-carbon
carbs -
What does it mean that glucose and fructose are isomers?
They have the same chemical formula but a different arrangement of atoms.
carbs isomers -
How is a disaccharide formed?
Two monosaccharides join via a glycosidic bond produced by a dehydration (condensation) reaction.
carbs reactions -
Give examples of polysaccharides and their functions.
- Starch: energy storage in plants
- Glycogen: energy storage in animals
- Cellulose: structural in plant cell walls
- Chitin: structural in exoskeletons and fungi cell walls
carbs polysaccharides -
proteins
-
proteins
-
What distinguishes the twenty amino acids from each other?
Each of the twenty amino acids is characterized by a unique 'R-group'.
proteins aminoacids -
Describe the basic parts of an amino acid (use the image for illustration).
An amino acid has an amino group (NH2), a carboxyl group (COOH), a central (alpha) carbon, a hydrogen, and an R-group.
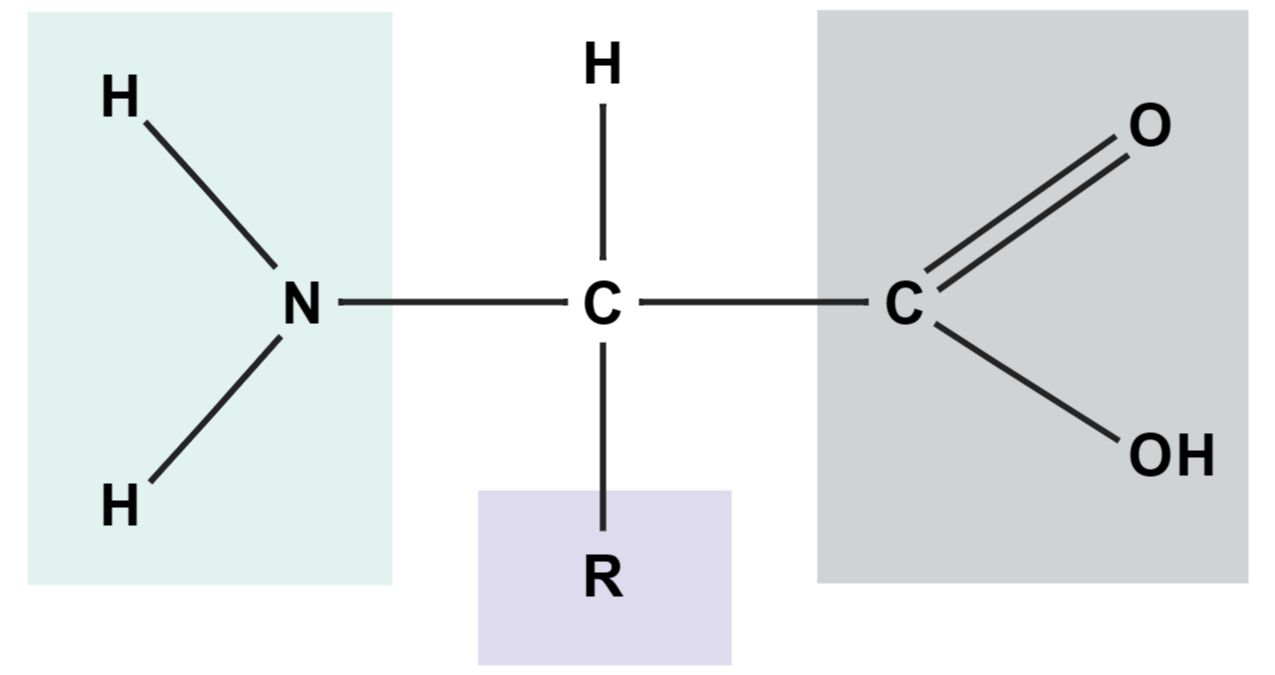 proteins aminoacids
proteins aminoacids -
What does the provided diagram illustrate about peptide formation?
The diagram shows two amino acids combining via a dehydration reaction to form a peptide bond with release of water.
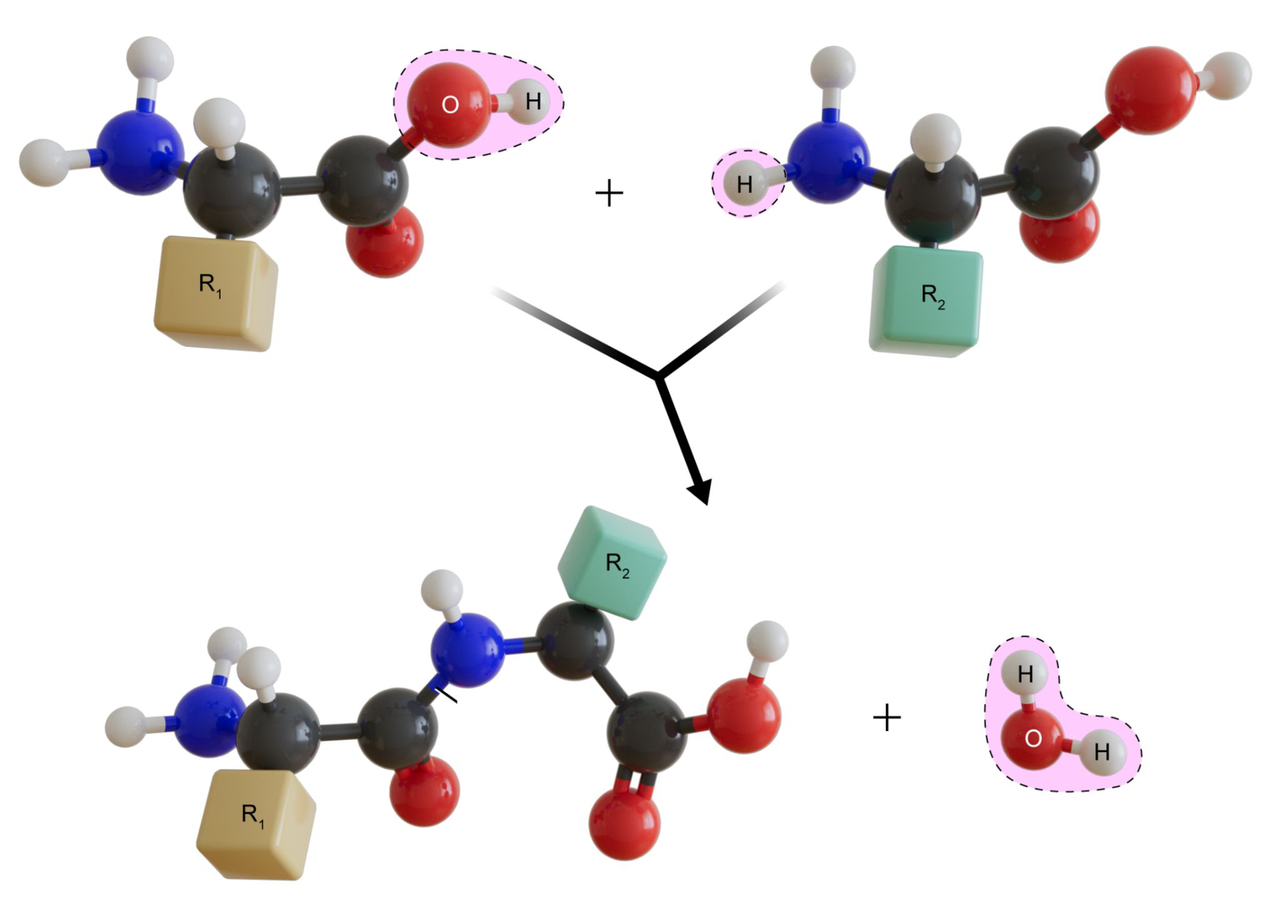 proteins peptide
proteins peptide -
protein polypeptide
-
How are peptide bonds formed and broken?
- Formed by dehydration (condensation) reactions
- Broken by hydrolysis reactions
peptide reactions -
What defines the primary structure of a protein?
The sequence of amino acids connected through peptide bonds.
protein primary -
What causes secondary protein structure and what shapes form?
Intermolecular hydrogen bonding between the polypeptide backbone (not R-groups) forming alpha-helices and beta-pleated sheets.
protein secondary -
What interactions stabilize tertiary protein structure?
Interactions between R-groups: hydrophobic interactions, disulfide covalent bonds between cysteines, hydrogen bonds, and ionic bonds.
protein tertiary -
What is quaternary protein structure?
Multiple polypeptide chains assembled together to form one functional protein.
protein quaternary -
What is protein denaturation and which structure remains intact?
Loss of protein function and higher order structures; only the primary structure remains unaffected.
denaturation protein -
Name factors that can cause protein denaturation.
- High or low temperatures
- pH changes
- Salt concentration changes
denaturation factors -
Give an example of denaturation from everyday life.
Cooking an egg: high heat disrupts intermolecular forces in egg proteins, causing coagulation.
denaturation example -
List common protein functions.
- Storage: reserve of amino acids
- Hormones: signaling molecules
- Receptors: bind signal molecules in membranes
- Structure: strength/support (hair, spider silk)
- Immunity: antibodies
- Enzymes: regulate reaction rates
protein functions -
How can the four levels of protein structure be visually illustrated?
Illustration of primary sequence, secondary alpha helix and beta sheet, tertiary 3D folding, and quaternary assembly.
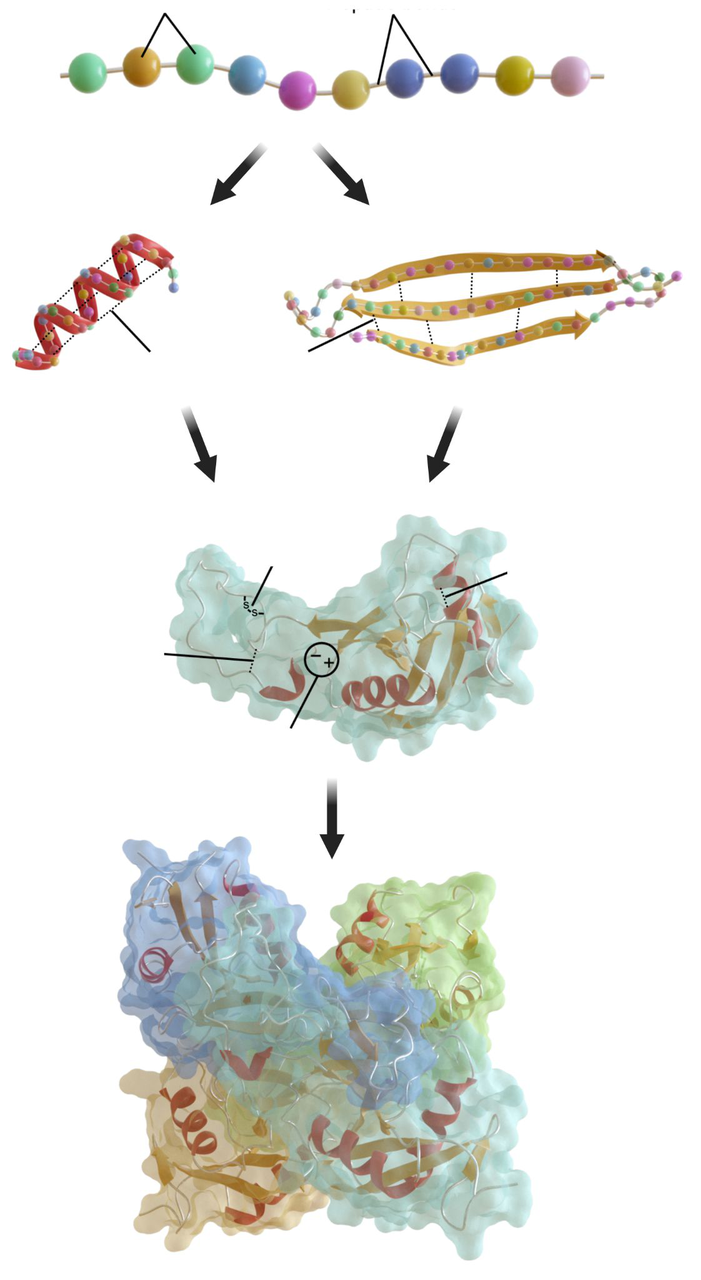 protein diagram
protein diagram -
What is the primary way catalysts increase reaction rates?
They lower the activation energy of a reaction.
catalysis chemistry -
What is the transition state in a chemical reaction?
The unstable conformation between the reactants and the products.
kinetics chemistry -
Do catalysts change reaction spontaneity or shift chemical equilibrium?
No — catalysts do not shift a chemical reaction or affect spontaneity.
catalysis thermodynamics -
enzymes biochemistry
-
Where do enzymes bind substrates and how specific is this site?
Enzymes bind substrates at an active site, which is specific for the substrate it acts upon.
enzymes structure -
What does the induced fit theory describe?
The active site molds and changes shape to fit the substrate when it binds.
enzymes mechanism -
enzymes rna
-
What is a cofactor and what is a coenzyme?
A cofactor is a non-protein molecule that helps enzymes; a coenzyme is an organic cofactor (e.g., vitamins).
enzymes cofactors -
cofactors metals
-
What makes protein enzymes susceptible to loss of function?
Protein enzymes are susceptible to denaturation and require optimal temperatures and pH for function.
enzymes stability -
Name mechanisms by which enzymes catalyze reactions.
- Conformational changes that bring reactive groups closer
- Presence of acidic or basic groups
- Induced fit
- Electrostatic attractions between enzyme and substrate
enzymes mechanism -
enzymes phosphatase
-
How does a phosphorylase add a phosphate group?
It directly adds a phosphate to a substrate by breaking bonds within a substrate molecule.
enzymes phosphorylase -
How does a kinase add a phosphate group to a substrate?
Kinases transfer a phosphate from an ATP molecule to a substrate and do not break substrate bonds to add the phosphate.
enzymes kinase -
What is feedback regulation of enzymes?
When the end product of an enzyme-catalyzed reaction inhibits the enzyme by binding to an allosteric site.
regulation enzymes -
How does competitive inhibition affect enzyme activity and how can it be overcome?
A competitive inhibitor competes with the substrate for the active site and can be outcompeted by adding more substrate.
inhibition competitive -
How does noncompetitive inhibition affect the enzyme and can it be outcompeted by more substrate?
A noncompetitive inhibitor binds an allosteric site, modifies the active site, and cannot be outcompeted by adding more substrate.
inhibition noncompetitive -
Which inhibition type is illustrated by an inhibitor binding an allosteric site and modifying the active site? (See illustration on answer)
Noncompetitive inhibition.
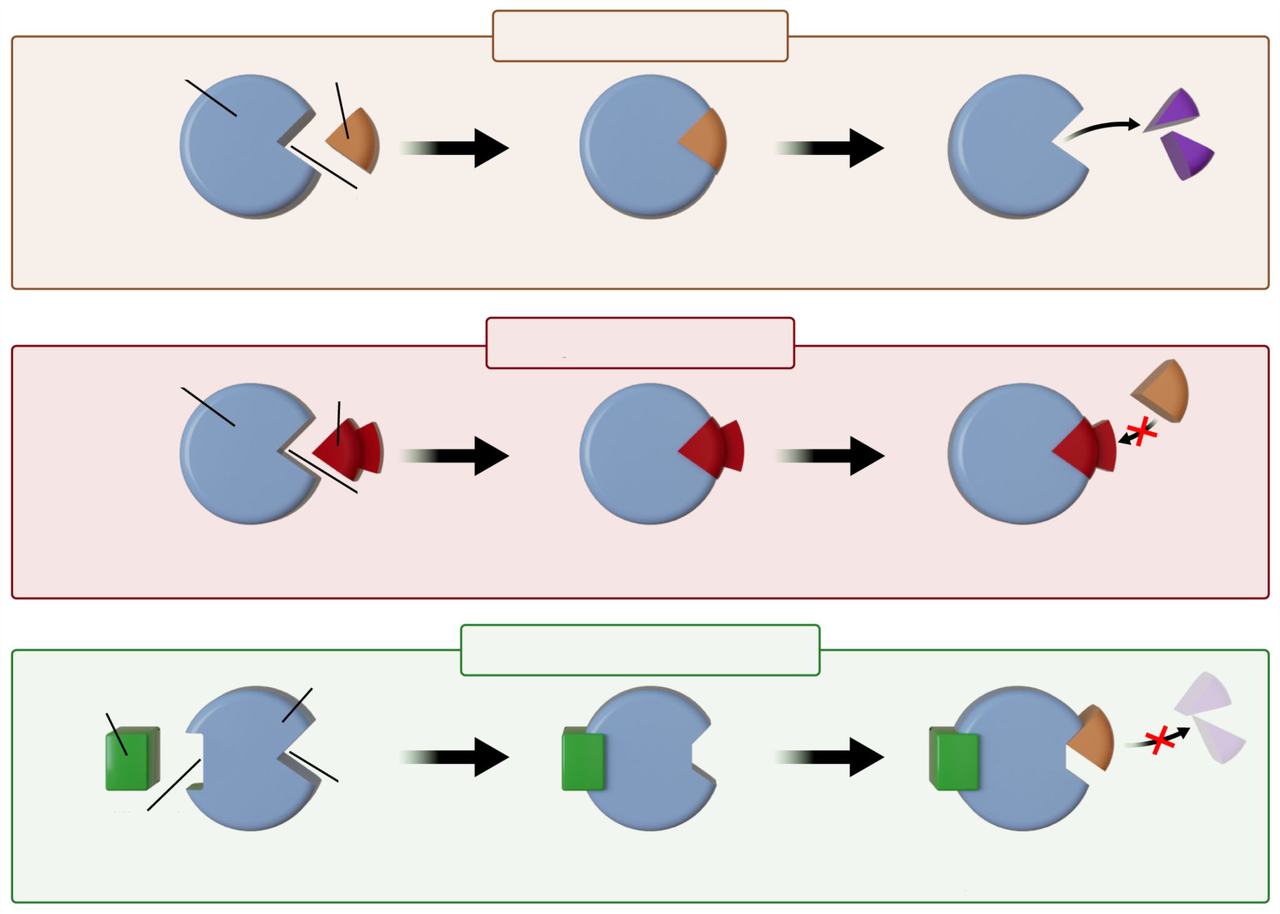 inhibition image
inhibition image -
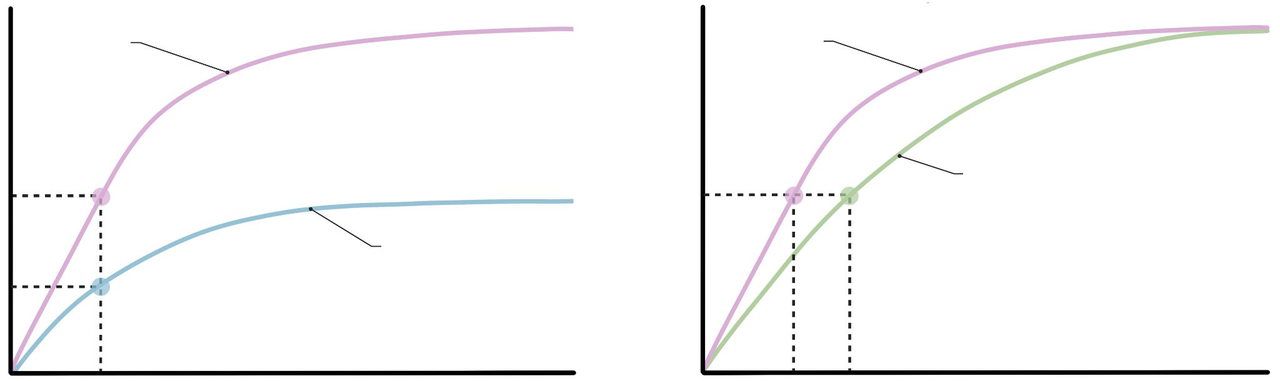
What do the axes represent on an enzyme kinetics plot of velocity versus substrate?
- x-axis: substrate concentration [X]
- y-axis: reaction rate or velocity (V)
enzyme kinetics plot -
What does the symbol V represent in the provided enzyme kinetics notes?
V is the maximum reaction velocity
enzyme kinetics vmax -
What is the Michaelis constant (K) in enzyme kinetics?
K is the substrate concentration [X] at which the velocity (V) is 50% of the maximum reaction velocity (Vmax)
enzyme kinetics km -
What is meant by saturation in an enzyme kinetics plot?
Saturation is when all active sites are occupied so the reaction rate plateaus despite increasing substrate concentration
enzyme kinetics saturation -
enzyme kinetics inhibition competitive
Chapter 1 — Molecules and Fundamentals of Biology
Quick outline
- Biological chemistry and macromolecules
- Carbohydrates: mono-, di-, polysaccharides and functions
- Proteins: amino acids, peptide bonds, 4 levels of structure, functions
- Enzymes: catalysis, cofactors, kinetics, inhibition
Biological chemistry — key terms
- Matter: anything with mass and volume.
- Element: pure substance that cannot be broken down chemically.
- Atom: smallest unit retaining element properties.
- Molecule: two or more atoms bonded.
- Intramolecular forces: forces within a molecule (e.g., covalent bonds).
- Intermolecular forces: forces between molecules (affect physical properties).
- Monomer / Polymer: monomers are single building blocks; polymers are chains of monomers.
- Dehydration (condensation): forms a covalent bond between monomers and releases water.
- Hydrolysis: breaks a covalent bond using water.
Carbohydrates — structure and roles
- Composed of C, H, O; main roles: fuel and structure.
- Classes:
- Monosaccharides: single sugars (examples: ribose — 5C; glucose and fructose — 6C).
- Disaccharides: two monosaccharides joined by a glycosidic bond (formed by dehydration). Examples: sucrose, lactose, maltose.
- Polysaccharides: long polymers of sugars. Examples and roles:
- Starch: plant energy storage.
- Glycogen: animal energy storage.
- Cellulose: plant structural polymer.
- Chitin: structural in exoskeletons and fungal cell walls.
- Note: glucose and fructose are isomers (same formula, different arrangement).
Proteins — building blocks and functions
Amino acids and peptide bonds
- Proteins are polymers of amino acids (contain C, H, O, N).
- Each amino acid has: amino group, carboxyl group, central (alpha) carbon, and an R-group (side chain).
- Amino acids join via peptide bonds formed by dehydration; hydrolysis breaks peptide bonds.


Protein structure levels
- Primary: linear amino acid sequence (peptide bonds).
- Secondary: local folding of backbone due to hydrogen bonds; forms alpha helices and beta sheets.
- Tertiary: 3D folding from interactions among R-groups (hydrophobic interactions, hydrogen bonds, ionic bonds, disulfide bridges between cysteines).
- Quaternary: assembly of multiple polypeptide chains into a functional protein.

Functions of proteins
- Storage: reserve amino acids.
- Hormones: signaling molecules.
- Receptors: membrane proteins that bind signals.
- Structural: provide strength/support (e.g., hair, spider silk).
- Immunity: antibodies.
- Enzymes: accelerate chemical reactions.
Denaturation
- Loss of higher-order structure (secondary, tertiary, quaternary) and function; primary sequence remains intact.
- Caused by extreme temperature, pH changes, salts; example: egg coagulation when cooked.
Enzymes and catalysis
- Catalysts lower activation energy and increase reaction rate but do not change reaction spontaneity or equilibrium.
- Enzymes are biological catalysts (mostly proteins); some RNAs (ribozymes) can be catalytic.
- Enzymes bind substrates at an active site; binding often follows the induced fit model (active site molds to substrate).
- Cofactors: non-protein helpers required for some enzymes; coenzymes are organic cofactors (e.g., vitamins); inorganic cofactors are often metal ions.
- Enzyme activity depends on optimal temperature and pH; enzymes can denature.
Mechanisms enzymes use to catalyze reactions
- Bring reactive groups into proximity or correct orientation.
- Provide acidic or basic side chains to facilitate reaction.
- Stabilize transition states (lower activation energy).
- Use electrostatic interactions to favor binding.
High-yield enzyme types
- Phosphatase: removes a phosphate group from a substrate.
- Phosphorylase: adds phosphate by breaking bonds within a substrate.
- Kinase: transfers a phosphate from ATP to a substrate (does not break substrate bonds to add phosphate).
Enzyme regulation and inhibition
-
Feedback inhibition: end product of a pathway inhibits an enzyme earlier in the pathway by binding an allosteric site.
-
Competitive inhibition:
- Inhibitor competes with substrate for active site.
- Effect can be reversed by increasing substrate concentration.
-
Observed kinetic change: apparent increase in \(K_m\), \(V_{max}\) unchanged.
-
Noncompetitive inhibition:
- Inhibitor binds an allosteric site, changing enzyme shape and activity.
- Cannot be overcome by increasing substrate.
- Observed kinetic change: \(V_{max}\) decreases, \(K_m\) unchanged.

Enzyme kinetics — essentials
- Plot axes: x = substrate concentration \([S]\), y = reaction velocity \(V\).
- \(V_{max}\): maximum reaction velocity when enzyme is saturated.
- Michaelis constant \(K_m\): substrate concentration at which \(V = \tfrac{1}{2}V_{max}\).
- Saturation: when increasing \([S]\) no longer increases rate because active sites are full.
Study tips and high-yield points
- Memorize monomer/polymer relationships for each macromolecule (sugars, amino acids, nucleotides, fatty acids).
- Remember the four levels of protein structure and what bonds/interactions stabilize each level.
- Practice interpreting how competitive vs noncompetitive inhibitors change \(K_m\) and \(V_{max}\).
- Understand dehydration vs hydrolysis reactions and be able to identify peptide and glycosidic bonds.