Meld je aan om meer functies te ontgrendelen
- Sla dit deck op in je account
- Oefen met flashcards via gespreide herhaling
- Exporteer naar Anki (.apkg) of PDF
- Verwerk documenten tot 100 pagina's
- Afbeeldingen uit PDF's en documenten gehaald
- Betere tekstherkenning uit je PDF's en documenten
- Betere flashcards dankzij ons geavanceerdere AI-model
Flashcards in dit deck (74)
-
How does the number of protons in the nucleus change across a period (left to right) in the periodic table?
The number of protons increases from left to right across a period.
periodic protons -
What is the effect on valence electrons when the number of protons in the nucleus increases across a period?
Increasing protons increases the positive electrostatic attraction of the nucleus acting on the valence shell.
electrostatics valence -
Which factor causes stronger attraction between nucleus and valence electrons across a period?
A higher nuclear positive charge due to more protons increases electrostatic attraction.
nuclear attraction -
Illustration: What concept does the diagram showing electrostatic lines of force between a nucleus and an electron illustrate?
It illustrates electrostatic attraction between the positively charged nucleus (protons) and an orbiting electron.
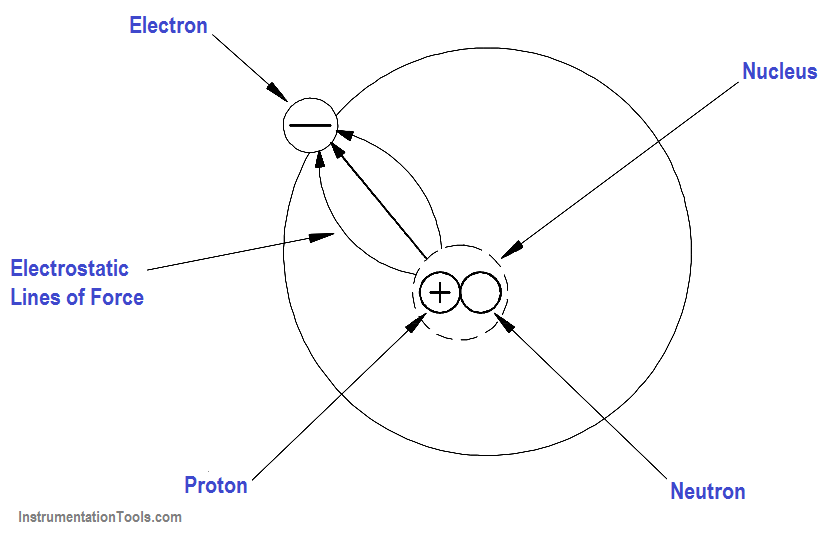 diagram illustration
diagram illustration -
What causes electrons to be attracted toward an atom's nucleus?
Electrons are attracted to the positively charged protons in the nucleus by electrostatic force.
electrostatics atoms -
How does the strength of electrostatic attraction affect the force on an electron?
The stronger the attraction, the greater the attractive force applied to the electron.
electrostatics force -
What is the effect of inner (core) electrons on outer (valence) electrons?
- Inner electrons 'shield' or reduce the nucleus's attractive forces on outer electrons.
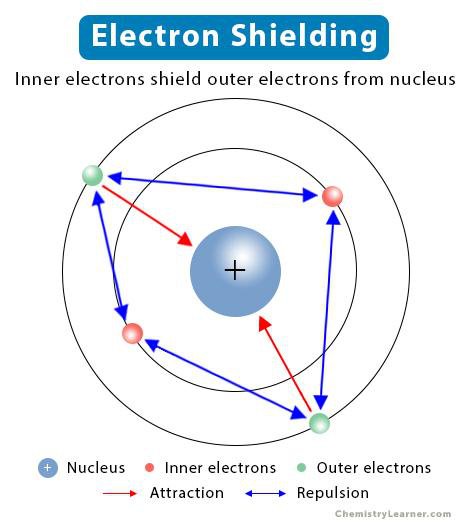 shielding periodicity
shielding periodicity -
What is core charge in atoms?
Core charge is a measure of the attractive force between valence electrons and the nucleus.
chemistry core_charge -
Why do valence electrons not feel the full positive charge of the nucleus?
Valence electrons do not feel the full positive charge because inner electrons act as a shield between them and the nucleus.
chemistry shielding -
What effect do inner electrons have on the nucleus' pull on valence electrons?
Inner electrons reduce the pull of the nucleus on valence electrons by shielding, lowering the effective attractive force.
chemistry shielding -
atomic radius
-
What two distances are illustrated in the atomic radius diagram?
- Atomic radius (nucleus to valence-shell electrons)
- Distance between nuclei (distance between the centers of two adjacent atoms)
diagram measurement -
Show the diagram that illustrates atomic radius.
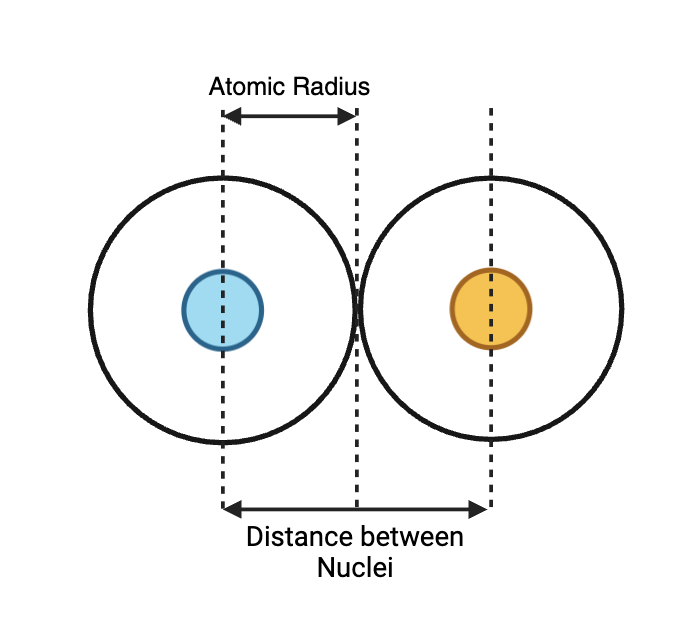
- Atomic radius: distance from nucleus to valence-shell electrons
diagram image -
What is the trend of atomic radius when moving across a period from left to right?
Atomic radius decreases across a period (left → right).
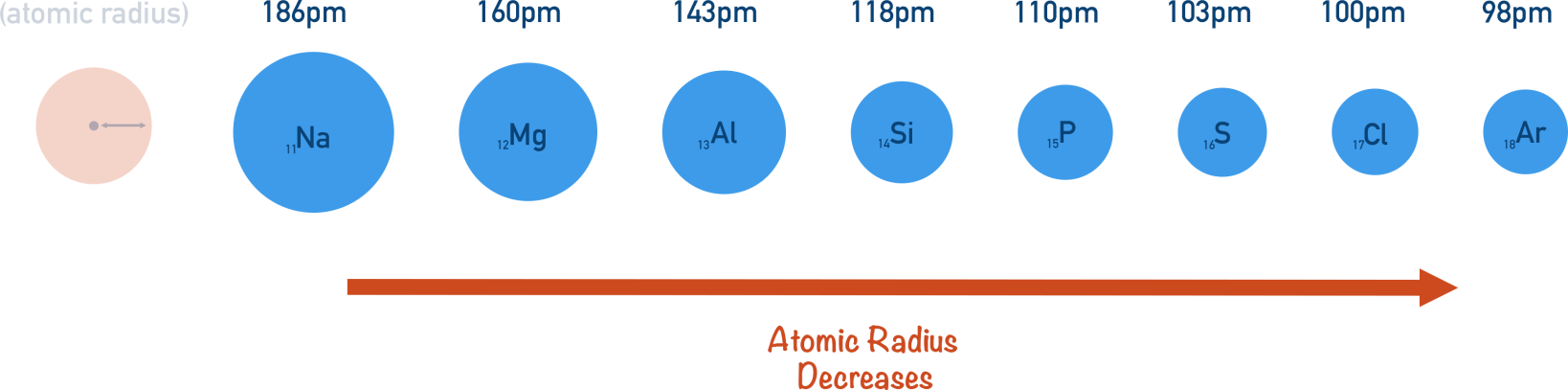 atomic-radius periodic-trends
atomic-radius periodic-trends -
Why does atomic radius decrease across a period?
Because the number of protons increases, increasing the positive nuclear charge that pulls valence electrons closer, reducing atomic size.
nuclear-charge atomic-radius -
Why does the atomic radius generally increase when moving down a group in the periodic table?
Because each step down adds an extra electron shell, placing valence electrons farther from the nucleus and increasing the atomic radius.
atomic-radius periodic-trends -
How does adding an extra electron shell affect valence electrons' attraction to the nucleus?
Valence electrons are further from the positive nucleus and therefore experience weaker electrostatic attraction.
shielding electrostatics -
What role do inner electron shells play in determining atomic radius down a group?
Inner shells shield valence electrons from the full positive charge of the nucleus, reducing effective attraction and increasing radius.
shielding atomic-radius -
Does nuclear charge increase or decrease down a group, and how does this compare to the effect of added shells?
Nuclear charge increases (more protons) down a group, but the effect is offset by added electron shells and shielding, so atomic radius still increases.
nuclear-charge periodic-trends -
In which direction does atomic radius change across a period?
Atomic radius increases from left to right across a period.
periodic_trends atomic_radius -
In which direction does atomic radius change down a group?
Atomic radius increases from top to bottom down a group.
periodic_trends atomic_radius -
Show an illustration of the atomic radius trend on the periodic table.
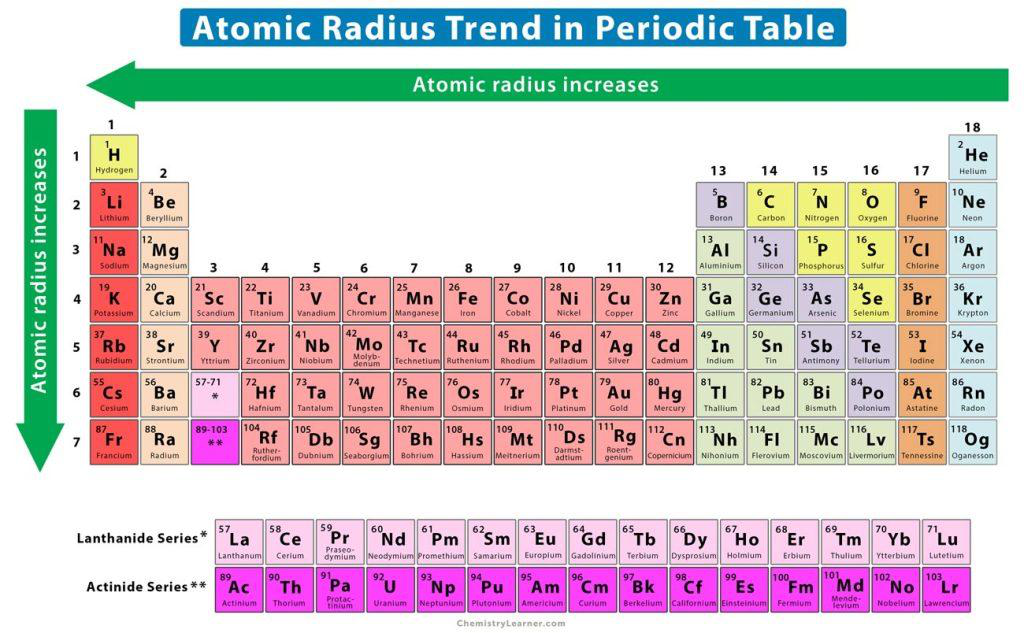
The diagram shows atomic radius increasing from left to right across a period and from top to bottom down a group.
periodic_trends diagram -
What is the first ionisation energy?
First ionisation energy is the energy required to remove one electron from each atom in 1 mole of gaseous atoms, forming 1 mole of gaseous ions.
ionisation energy periodic -
What occurs when an atom in the gaseous state undergoes ionization?
- The atom loses an electron and becomes a positively charged ion (A → A+ + e−)
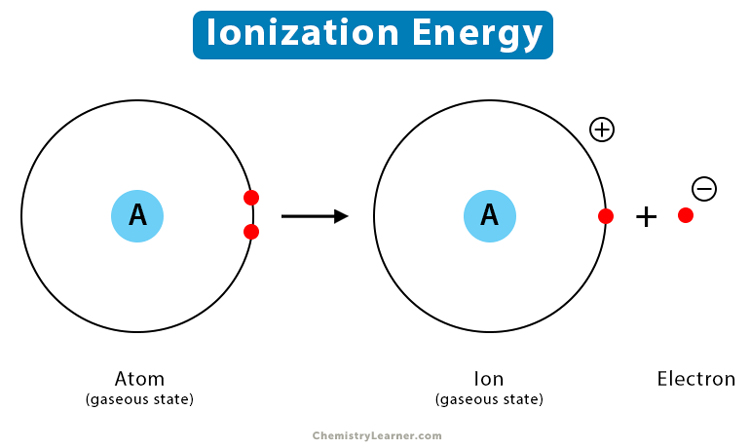 ionisation process diagram
ionisation process diagram -
What is the trend in first ionisation energy across a period (left → right)?
First ionisation energy increases across a period (left → right).
ionisation periodicity -
How does core charge change across a period, and what stays the same?
Across a period the core charge increases (more protons) while the number of electron shells stays the same.
corecharge electronicstructure -
Why does increasing core charge raise first ionisation energy across a period?
Increased core charge causes stronger electrostatic attraction between the nucleus and valence electrons, so more energy is needed to remove an electron.
electrostatic ionisation -
Which region of the periodic table contains elements with high first ionisation energies?
Elements on the right of the periodic table (for example Ne and Ar) have high first ionisation energies.
examples periodictable -
What is the trend of first ionisation energy down a group (top → bottom)?
First ionisation energy decreases down a group.
ionisation periodic -
How does having more electron shells (down a group) affect valence electrons?
More electron shells place valence electrons further from the nucleus.
atomic radius -
What is the effect of inner-shell electron shielding on ionisation energy?
Greater shielding from inner-shell electrons reduces electrostatic attraction, lowering ionisation energy.
shielding ionisation -
Why is less energy required to remove an electron from elements lower in a group?
Weaker electrostatic attraction between nucleus and valence electron means less energy is required to remove an electron.
electrostatic ionisation -
Give two examples of elements at the bottom of a group that have very low first ionisation energies.
- Cs
- Fr
examples ionisation -
What general trend is shown for ionization energy in the periodic table?
Ionization energy increases.
ionization trend -
According to the periodic table image, in which directions does ionization energy increase?
- Across a period: from right to left
- Down a group: from top to bottom
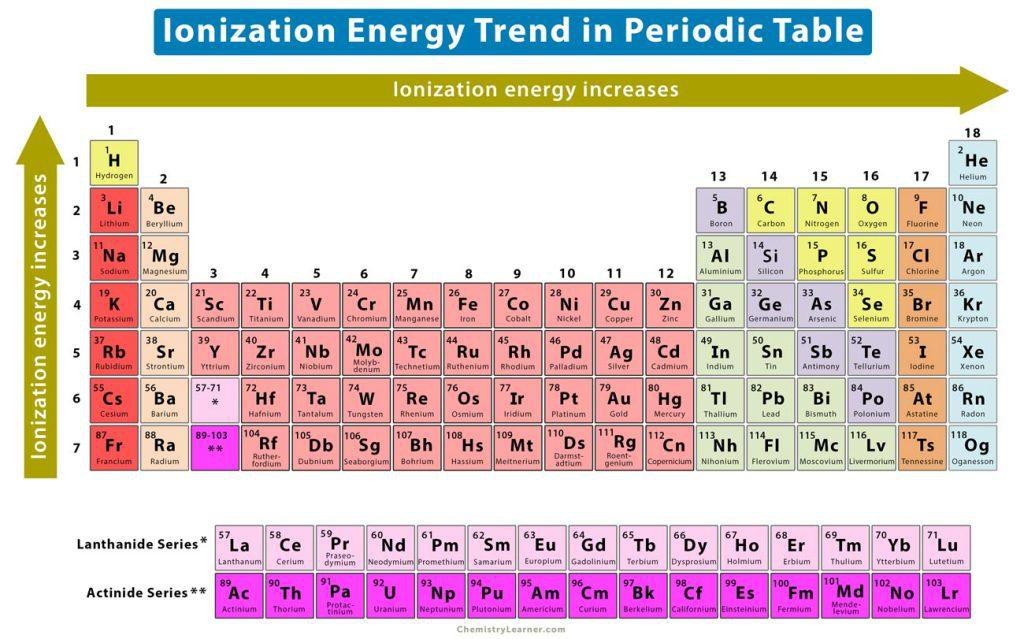 ionization periodic-trends image
ionization periodic-trends image -
What is electronegativity?
Electronegativity is the ability of an atom to attract electrons towards itself when it is involved in a chemical bond.
electronegativity chemistry -
In a covalent bond, where are the shared electrons located relative to the more electronegative atom?
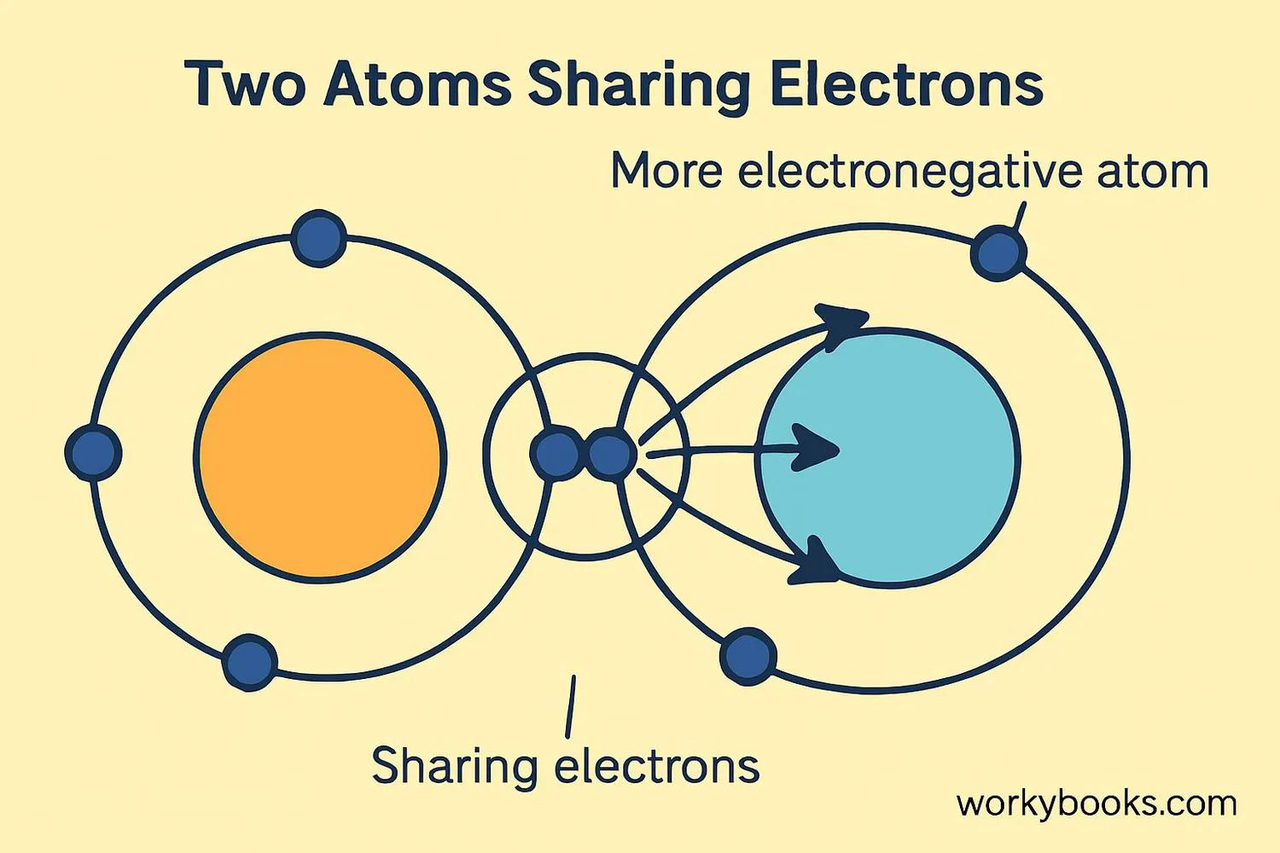
The shared electrons are closer to the more electronegative atom.
bonding electronegativity -
How does electronegativity change across a period (left → right)?
Electronegativity increases across a period (left → right).
electronegativity periodic-trends -
What happens to nuclear charge across a period?
The number of protons (nuclear charge) increases across a period.
nuclear-charge periodic-trends -
Why is shielding similar for valence electrons across a period?
Valence electrons are in the same shell, so shielding by inner electrons is similar across a period.
shielding electron-configuration -
What is the effect of increased nuclear charge with similar shielding on bonding electrons?
There is a stronger attraction between the nucleus and bonding electrons.
electrostatic-attraction bonding -
Where are the most electronegative elements located on the periodic table and give examples?
Most electronegative elements are on the top right of the periodic table; examples: F, O, N.
periodic-table examples -
How does electronegativity change down a group (top → bottom)?
Electronegativity decreases down a group.
electronegativity periodicity -
What happens to the number of electron shells as you move down a group?
Atoms have more electron shells moving down a group.
atomic structure -
How does an increased number of electron shells affect the distance between nucleus and bonding electrons?
More electron shells increase the distance between the nucleus and bonding electrons.
shielding distance -
What effect does increased shielding have on nuclear attraction for bonding electrons?
More shielding reduces nuclear attraction for bonding electrons.
shielding attraction -
Which elements in a group are the least electronegative?
Bottom-group elements are the least electronegative.
electronegativity groups -
chemistry electronegativity
-
chemistry noblegases
-
chemistry noblegases
-
Refer to the periodic table image: How does electronegativity change across a period?
Electronegativity increases from right to left across a period.
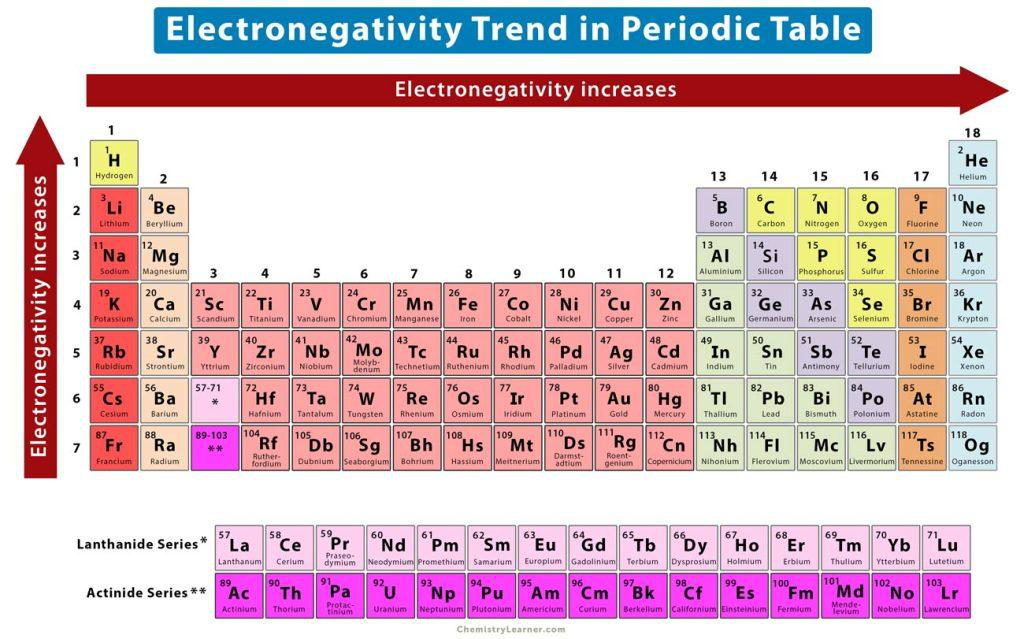 electronegativity periodic
electronegativity periodic -
How does electronegativity change down a group in the periodic table?
Electronegativity increases from top to bottom down a group.
electronegativity periodic -
What is metallic character?
Metallic character: How closely an element exhibits properties of a metal — how readily it loses an electron to form an ion.
chemistry periodic -
Name common properties of metals.
- Form cations
- Metallic luster
- Good conductors
- Malleable and ductile
- High melting point
- Mostly solid at room temperature
- Sonorous
 chemistry metals
chemistry metals -
Name common properties of nonmetals.
- Form anions
- Dull, colorless to colorful
- Poor conductors
- Brittle
- Low melting point
- Often liquids or gases at room temperature
- Not sonorous
chemistry nonmetals -
What happens to metallic character across a period from left to right?
Metallic character decreases across a period from left to right.
periodicity metallic -
How does nuclear attraction for electrons change as atomic number increases across a period?
Nuclei attract electrons more strongly as atomic number increases across a period.
periodicity nuclear -
Why do atoms become less metallic across a period?
Because stronger nuclear attraction makes it harder for atoms to lose electrons, so they are less metallic.
metallic ionisation -
Give an example sequence across a period showing decreasing metallic character.
- Na (very metallic)
- Mg
- Al
- Si
- P (non-metal)
examples periodicity -
What happens to metallic character of elements when moving down a group in the periodic table?
Metallic character increases when moving down a group.
periodicity metallic -
How does the number of electron shells change when moving down a group?
There are more electron shells when moving down a group.
electronic shells -
Why does having more electron shells increase metallic character?
More shells put valence electrons further from the nucleus, reducing attraction and increasing metallic character.
shielding metallic -
What is the effect of weaker attraction between nucleus and valence electrons on an element's behavior?
Weaker attraction means valence electrons are more easily lost, which increases metallic character.
ionisation metallic -
Give the example order of metallic character for Group 1 elements from least to most metallic.
- Li (least metallic)
- Na
- K
- Rb
- Cs (most metallic)
examples group1 -
In the periodic table, which direction across a period does metallic character increase?
From right to left across a period.
periodic_trends metallic -
What two opposite trends across a period are shown in the periodic-table image below?
- Metallic character increases from right to left
- Non-metallic character increases from left to right
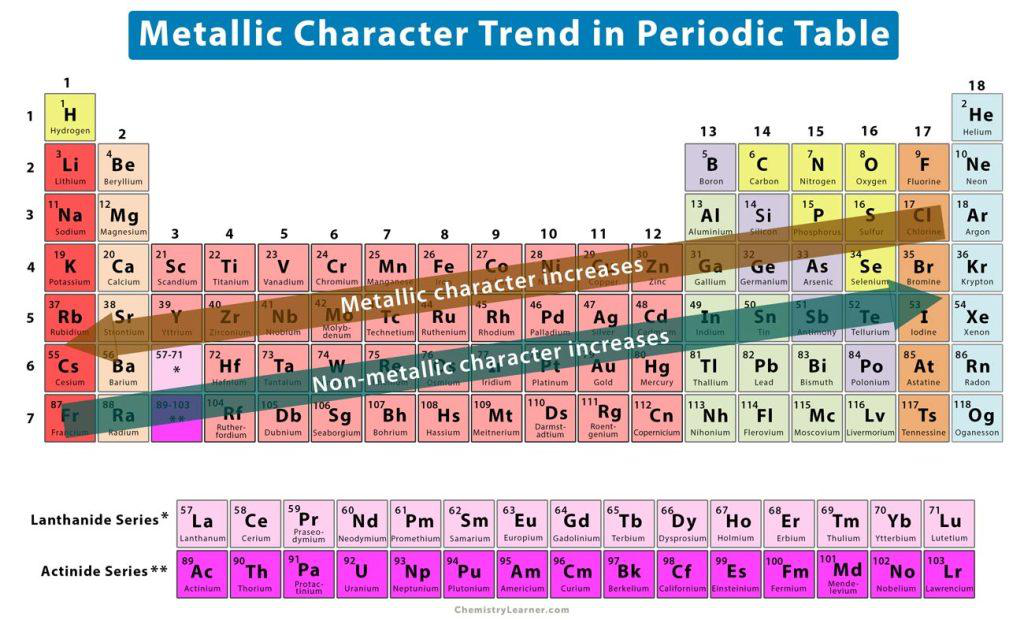 periodic_trends metallic nonmetallic
periodic_trends metallic nonmetallic -
In the periodic table, which direction does electronegativity increase?
- Left to right and bottom to top
electronegativity trends -
atomic_radius trends
-
Which direction does metallic character increase on the periodic table?
- Diagonally from top-left to bottom-right
metallic trends -
Which direction does non-metallic character increase on the periodic table?
- Diagonally from bottom-right to top-left
nonmetallic trends -
Which direction do ionization energy and electron affinity increase on the periodic table?
- Diagonally from bottom-left to top-right
ionization_energy electron_affinity trends -
Name two special series of elements shown in the periodic trends overview.
- Lanthanide Series
- Actinide Series
lanthanides actinides series -
Referencing a diagram of periodic trends, what three features are identified in the legend?
- Alkali metals
- Alkaline earth metals
- Transition metals
legend groups periodic_table -
Where can a diagram summarizing periodic trends (electronegativity, atomic radius, metallic character, non-metallic character, ionization energy, electron affinity) be found?
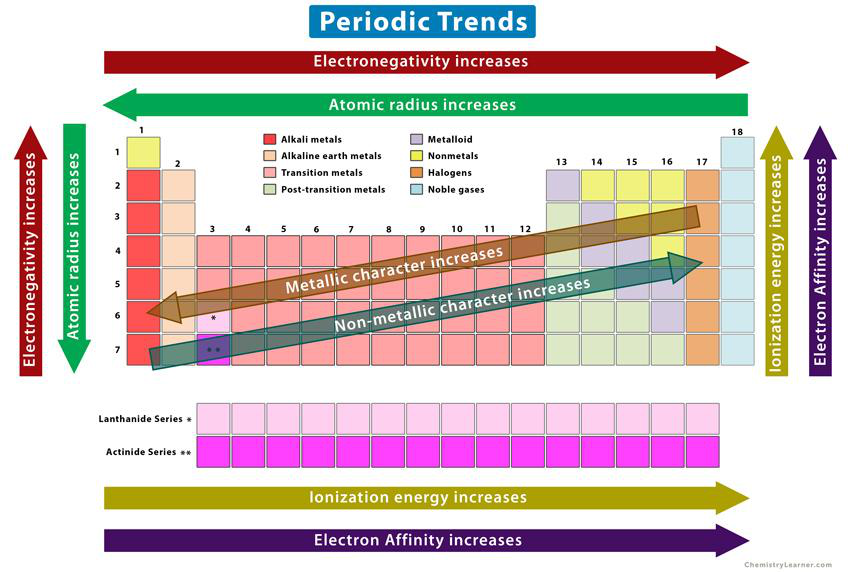 diagram visual
diagram visual
Periodic Trends — concise study notes
Quick overview
- Periodic trends arise from two main factors: nuclear charge (number of protons) and electron shielding (inner electrons reducing nuclear pull). These determine atomic radius, ionization energy, electronegativity, and metallic character.
Electrostatic attraction & shielding
- Electrostatic attraction: valence electrons are attracted to the positively charged nucleus; stronger nuclear charge → stronger attraction.
 Alt text: Electrostatic attraction between nucleus and electron.
Alt text: Electrostatic attraction between nucleus and electron.
- Electron shielding: inner-shell electrons repel and partly block the nucleus from valence electrons, reducing the net pull those electrons feel.
 Alt text: Electron shielding by inner-shell electrons.
Alt text: Electron shielding by inner-shell electrons.
- Effective nuclear charge (core charge): the net positive charge felt by valence electrons; often approximated as \(\(Z_{\text{eff}} \approx Z - S\)\) where \(Z\) is proton number and \(S\) is shielding by inner electrons.
Atomic radius
- Definition: the distance from the nucleus to the valence-shell boundary (often measured as half the distance between nuclei in a bond).
 Alt text: Atomic radius measured from nucleus to valence shell.
Alt text: Atomic radius measured from nucleus to valence shell.
Trend across a period (left → right)
- Atomic radius decreases because proton number increases while electron shell count stays the same, so valence electrons are pulled in more tightly.
Trend down a group (top → bottom)
- Atomic radius increases because each row adds an electron shell; valence electrons are farther out and more shielded.
First ionisation energy
- Definition: energy required to remove one electron from each atom in 1 mole of gaseous atoms, forming 1 mole of gaseous ions. \(\(A(g) \rightarrow A^{+}(g) + e^{-}\)\)
- Units commonly reported as kJ·mol^{-1}.
 Alt text: Atom in gas phase losing an electron to form a gaseous ion.
Alt text: Atom in gas phase losing an electron to form a gaseous ion.
Trend across a period (left → right)
- Ionisation energy increases because higher core charge (more protons) holds valence electrons more tightly.
Trend down a group (top → bottom)
- Ionisation energy decreases because valence electrons are farther out and more shielded, so less energy is needed to remove one.
Electronegativity
- Definition: an atom's tendency to attract bonding electrons in a chemical bond.
 Alt text: Shared electrons closer to the more electronegative atom.
Alt text: Shared electrons closer to the more electronegative atom.
Trend across a period (left → right)
- Electronegativity increases because nuclear charge rises while shielding remains similar, so bonded electrons are attracted more strongly.
Trend down a group (top → bottom)
- Electronegativity decreases because added shells increase distance and shielding.
Noble gases
- Noble gases typically have no electronegativity value since they rarely form covalent bonds with shared pairs.
Metallic character
- Definition: how readily an element loses electrons (forms cations) — metals lose electrons easily.
 Alt text: Comparison chart of metals and nonmetals properties.
Alt text: Comparison chart of metals and nonmetals properties.
Trend across a period (left → right)
- Metallic character decreases because atoms hold electrons more tightly and become less willing to lose them.
Trend down a group (top → bottom)
- Metallic character increases because outer electrons are farther from the nucleus and easier to remove.
Summary periodic map (quick reference)
- Atomic radius: decreases → across period, increases ↓ down group.
- Ionisation energy and electronegativity: increase → across period, decrease ↓ down group.
- Metallic character: decreases → across period, increases ↓ down group.
 Alt text: Periodic table showing directions of major chemical trends.
Alt text: Periodic table showing directions of major chemical trends.
Quick examples to remember
- Fluorine (top-right) — very high electronegativity and high ionisation energy, very small atomic radius.
- Cesium/Francium (bottom-left) — large atomic radius, low ionisation energy, highly metallic.
Study tips
- Link each trend to the two causes: proton number (nuclear charge) and shielding/number of shells.
- Practice by predicting properties for a given element (e.g., compare Na vs K, or O vs F).
- Use the periodic map image as a mnemonic: arrows show directions of increase.