Sign up to unlock more features
- Save this deck to your account
- Study flashcards with spaced repetition
- Export to Anki (.apkg) or PDF
- Process documents up to 100 pages
- Images extracted from PDFs and documents
- Better text extraction from your PDFs and documents
- Better flashcards with our more advanced AI model
Flashcards in this deck (24)
-
physics vaporization
-
physics vaporization
-
physics law_of_laplace
-
According to Dalton's Law, the total pressure in a mixture of gases equals the sum of the individual pressures.
physics dalton's_law -
physics poiseuille's_law
-
physics fluid_dynamics
-
physics fluid_dynamics
-
The formula for flow rate, according to Poiseuille's Law, is given by \(\(Q = \frac{\pi P r^4}{8\eta L}\)\).
physics poiseuille's_law -
According to Henry's law, the partial pressure of an anesthetic agent in the blood is proportional to the partial pressure of the volatile in the alveoli.
gas_laws henry's_law -
gas_laws o2
-
gas_laws charles_law
-
gas_laws gay-lussac's_law
-
Boyle's Gas Law describes that pressure and volume are inversely proportional at constant temperature.
gas_laws boyle's_law -
Fick's Law of Diffusion states that the rate of diffusion is directly proportional to partial pressure gradient, membrane area, and solubility of gas.
diffusion fick's_law -
According to Fick's Law, diffusion is indirectly proportional to membrane thickness and the square root of molecular weight.
diffusion fick's_law -
Diffusion hypoxia occurs due to high inspired % of nitrous oxide leading to hypoventilation and displacement of O2.
diffusion gas_laws -
In the mnemonic 'Could These Girls Possibly Be Virgins', each letter represents a different gas law including Charles Law and Boyle's law.
mnemonic gas_laws -
The components of the High Pressure system in an anesthesia machine include: - Hanger yoke - Yoke block with check valves - Cylinder pressure gauge - Cylinder pressure regulators.
anesthesia machine pressure -
The Intermediate Pressure system in an anesthesia machine starts at the pipeline and ends at the flowmeter valves.
anesthesia machine pressure -
Components of the Intermediate Pressure system include: - Pipeline inlets - Pressure gauges - Oxygen pressure failure device - Oxygen second stage regulator - Oxygen flush valve - Ventilator power inlet - Flowmeter valves.
anesthesia machine pressure -
anesthesia machine pressure
-
Components of the Low Pressure system include: - Flowmeter tubes (Thorpe tubes) - Vaporizers - Check valve (if present) - Common gas outlet.
anesthesia machine pressure -
The components of the anesthesia machine are divided into three pressure systems: High Pressure, Intermediate Pressure, and Low Pressure.
anesthesia machine pressure -
anesthesia machine overview
MODULE 1: PHYSICS / EQUIP & MONITORING / NMJ & MUSCLE RELAXANTS
PHYSICS
I. Vaporization
Vapor Pressures at 20°C
| Anesthetic Agent | Vapor Pressure (mmHg) |
|---|---|
| Sevoflurane | 170 |
| Isoflurane | 240 |
| Enflurane | 172 |
| Halothane | 244 |
| Desflurane | 669 |
- Vapor Pressure (VP): Increases with temperature; Desflurane vaporizer operates at ~2 atm (approx. 1500 mmHg).
- High-Low Relationships:
- H-L-H: Higher VP agent in lower VP vaporizer results in higher delivered concentration.
- L-H-L: Lower VP agent in higher VP vaporizer results in lower delivered concentration.
- Latent Heat of Vaporization: Energy lost during vaporization without temperature change.
II. Pressure & Tension
- Pressure Conversions:
- 1 mmHg = 1.36 cmH2O, 1 atm = 760 mmHg = 101 kPa
- Law of Laplace: Tension (\(T\)) on wall of a cylinder is proportional to its radius (\(r\)): \(\(T = P imes r\)\).
- Dalton's Law: Total pressure is the sum of individual gas pressures.
III. Fluid Dynamics
Poiseuille's Law
-
Describes how flow rate (\(Q\)) is influenced by radius (\(r\)), pressure (\(P\)), viscosity ($ ext{η}\(), and length (\)L$). \(\(Q = rac{ ext{π}Pr^4}{8 ext{η}L}\)\).
-
Laminar Flow: Flow determined by viscosity; to increase flow:
- Decrease needle gauge
- Shorten needle
- Raise IV pole.
- Turbulent Flow: Flow determined by density; helium used with O2 to decrease turbulence during upper airway obstruction.
IV. Solubility of Gases
- Henry's Law: Partial pressure of an anesthetic agent in blood is proportional to its alveolar pressure. Calculation example for dissolved gases in blood:
- Dissolved O2: 0.003 mL/100mL/mmHg
- Dissolved CO2: 0.067 mL/100mL/mmHg.
V. Gas Laws
- Charles's Law: \(V_1/T_1 = V_2/T_2\); volume and temperature are directly proportional at constant pressure.
- Gay-Lussac's Law: \(P/T = K\); pressure and temperature are directly proportional at constant volume.
- Boyle's Law: \(PV = K\); pressure and volume are inversely proportional at constant temperature.
- Fick's Law of Diffusion: Rate of diffusion is directly proportional to partial pressure gradient, membrane area, and gas solubility; inversely proportional to thickness and square root of molecular weight.
VI. Equipment & Monitoring
Anesthesia Machine Overview
- High, Intermediate, and Low Pressure systems:
| Pressure System | Location | Components |
|---|---|---|
| High Pressure | Begins at the cylinder | Cylinder gauges, regulators |
| Intermediate | Pipeline to flowmeter | Pressure gauges, flowmeter valves |
| Low Pressure | Flowmeter to gas outlet | Vaporizers, common gas outlet |
Additional Visuals
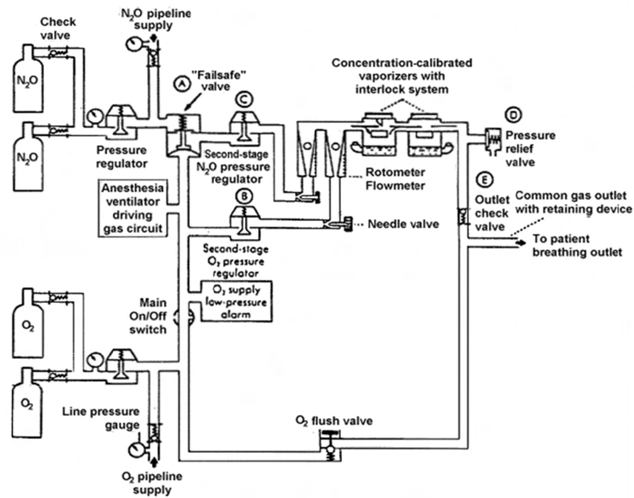 - Provides component overview of an anesthesia machine.
- Provides component overview of an anesthesia machine.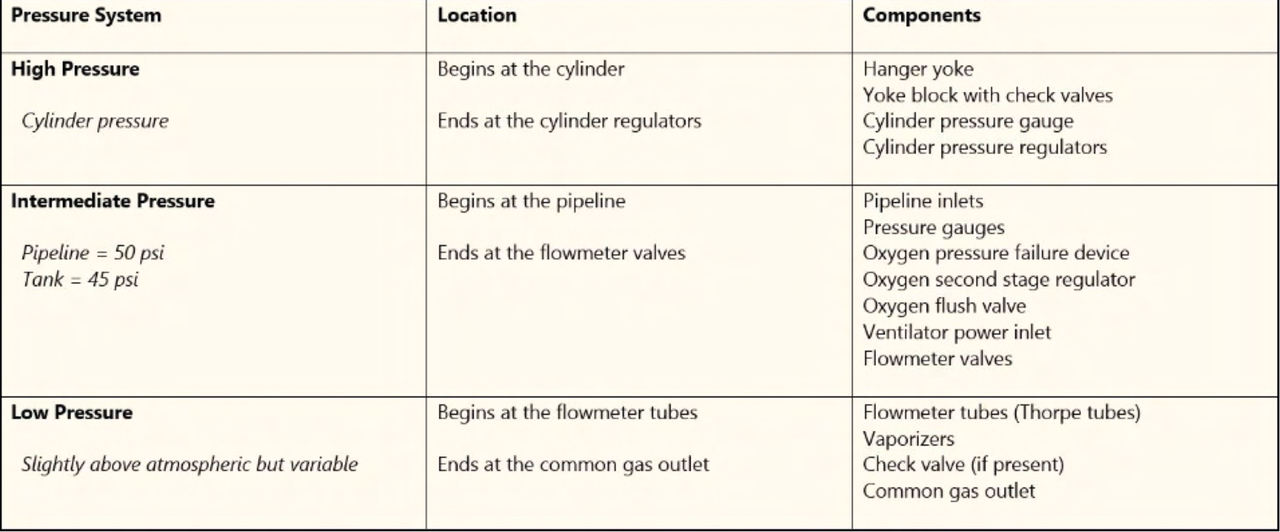 - Describes different pressure systems in anesthesia machines.
- Describes different pressure systems in anesthesia machines.
Memory Tasks
- Essential readings from key sources.
- Key formulas and their applications in clinical practice.
- Understanding anesthesia machine components and functionalities.