Sign up to unlock more features
- Save this deck to your account
- Study flashcards with spaced repetition
- Export to Anki (.apkg) or PDF
- Process documents up to 100 pages
- Images extracted from PDFs and documents
- Better text extraction from your PDFs and documents
- Better flashcards with our more advanced AI model
Flashcards in this deck (39)
-
What is the definition of glass?
A hard, brittle material typically made by heating silica sand with soda and lime.
properties definition -
What is Glass Transition Temperature (Tg)?
The temperature at which glass transitions from a hard and brittle state to a plastic state.
properties tg -
types glass
-
applications industries
-
properties glass
-
Identify a characteristic of broken glass.
It is fragmented into many small, irregular shapes held together in a network.
characteristic glass -
How is tempered glass different from regular glass?
It is treated thermally to increase its strength and thermal resistance.
types glass -
materials properties
-
Define glass in terms of its structure.
An amorphous solid that lacks a long-range ordered structure.
materials definition -
What is the Glass Transition Temperature (Tg)?
The temperature range where glass transitions from a brittle state to a malleable state.
materials thermodynamics -
List the types of glass and their general uses.
- Float glass: Windows
- Borosilicate glass: Labware
- Soda-lime glass: Containers
materials types -
materials definition
-
materials structure
-
Define the Glass Transition Temperature (Tg).
The temperature at which glass transitions from a hard to a rubbery state.
materials thermal_properties -
What factors determine the composition of glass?
- Silica content
- Additives (like soda and lime)
- Intended application
materials composition -
What kind of structure does glass have?
Non-crystalline (vitreous) or amorphous structure with molecules in random orientations.
structure glass -
Do the molecules in glass have long-range order?
No, they are not arranged in a regular repetitive long-range order.
molecules glass -
molecules structure
-
materials glass
-
What type of materials do not solidify like crystalline materials?
Glassy or non-crystalline materials
glass materials -
How does a glass behave when cooled?
It becomes more viscous continuously with decreasing temperature.
glass properties -
Above the glass transition temperature, what state is glass in?
First a super-cooled liquid, then a liquid.
materials glass states -
How does the volume of crystalline materials behave as they solidify?
Solidify sharply at the melting temperature (Tm).
materials crystallization -
What defines the non-crystalline state of a material?
The characteristic glass transition temperature, Tg.
materials glass -
What defines the Glass Transition Temperature (Tg)?
The point of intersection of the two slopes in the specific volume vs. temperature curve.
glass tg -
What effect do faster cooling rates have on Tg?
They produce higher values of the Glass Transition Temperature (Tg).
glass properties -
glass properties
-
glass viscosity melting_point
-
glass types applications
-
How do alkaline earth oxides like CaO affect glass?
They help reduce viscosity and improve workability.
materials glass -
What is a characteristic of modified glass like soda-lime glass?
It has a network where Na+ does not integrate into the structure.
glass types -
What is a characteristic of intermediate oxide glass?
It includes Al3+ cations that are integrated into the glass network.
glass types -
What is the melting temperature characteristic of fused silica?
Very high melting temperature, shock resistant.
glass fused_silica -
glass soda_lime
-
glass fiberglass
-
What is the most commonly produced glass?
Soda-lime glass accounts for about 90% of all glasses produced.
glass composition -
What are some uses of soda-lime glass?
- Flat glass
- Containers
- Pressed and blown ware
- Lighting products
glass applications -
materials advantages
-
Why are lead glasses used for some optical and decorative glasses?
They have a high refractive index
materials optics
Introduction to Glasses
GLASSES
This lecture covers: * Properties of glasses and their significance * Definitions and structure of glasses * Glass Transition Temperature (Tg) * Glass compositions * Conclusions
Properties of Glasses
PROPERTIES OF GLASSES
- Transparent and hard at room temperature
- Strong and corrosion-resistant
- Essential in construction and automotive industries
- Important for electrical components like lamps

- Key for telecommunications via optical fibers

Definition of Glass
DEFINITION OF A GLASS
- Glass is an inorganic ceramic made from oxides, fused and cooled without crystallization.
- Defined as an inorganic (ceramic) product of fusion that solidifies without crystallization.
- Exhibits a non-crystalline (vitreous) or amorphous structure with random molecular orientation.
Structure of Glasses
STRUCTURE OF GLASSES
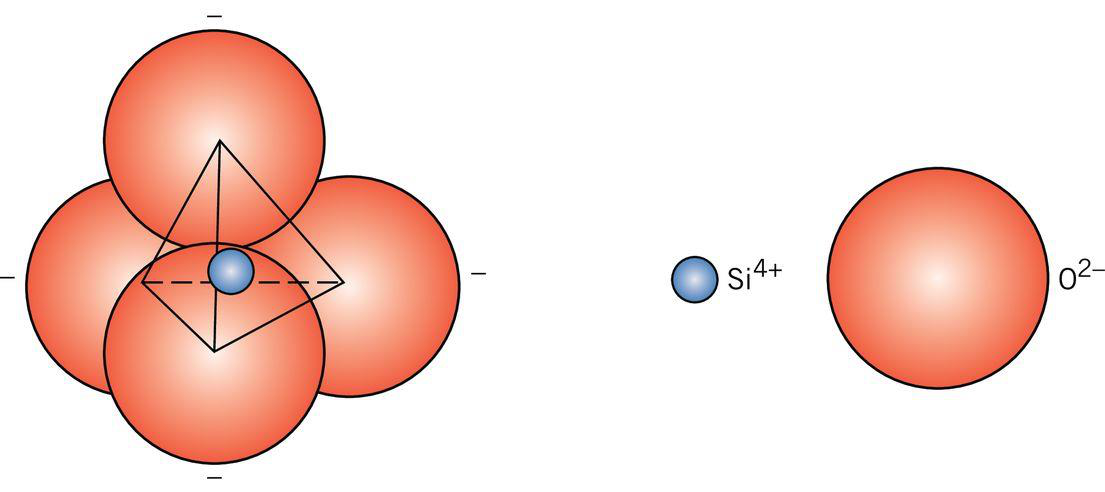
- Typically based on silica (SiO₂) with SiO4 tetrahedra as fundamental units.
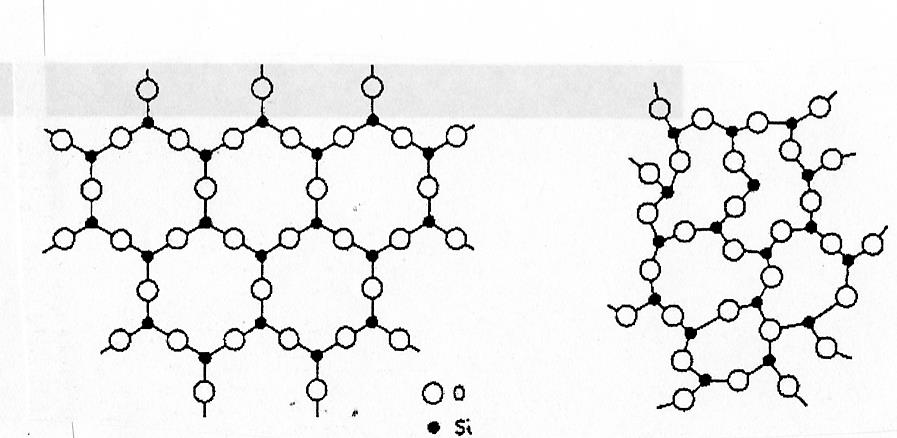
- Difference in arrangement between crystalline and amorphous states:
- Crystalline: long-range order (e.g., cristobalite)
- Amorphous: loose network without long-range order
Glass Transition Temperature
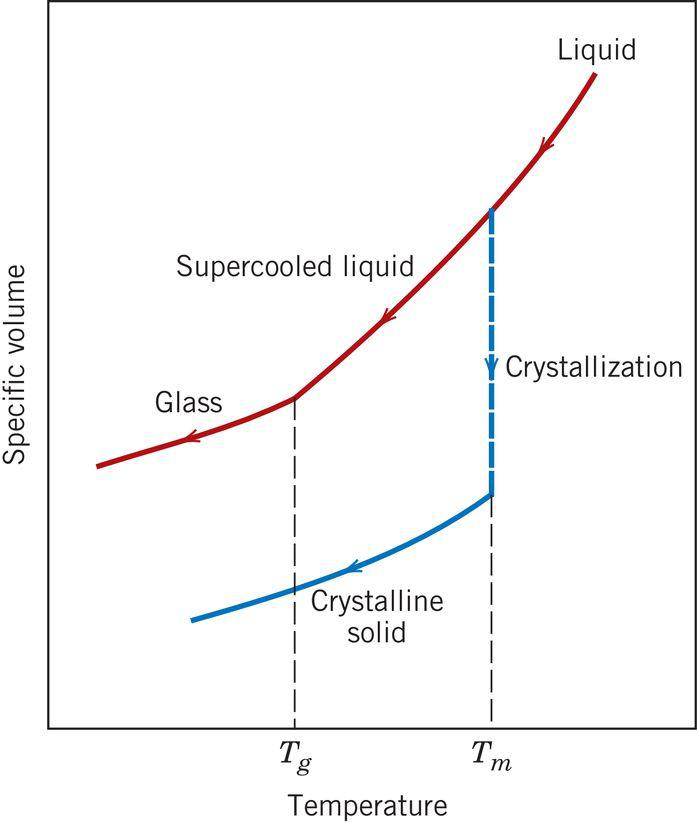
COOLING OF GLASS
- Glass does not solidify like crystalline materials; it gradually becomes viscous upon cooling.
- Defines Tg: glass transition temperature, below which glass is rigid and above which it's a supercooled liquid.
Viscosity Characteristics
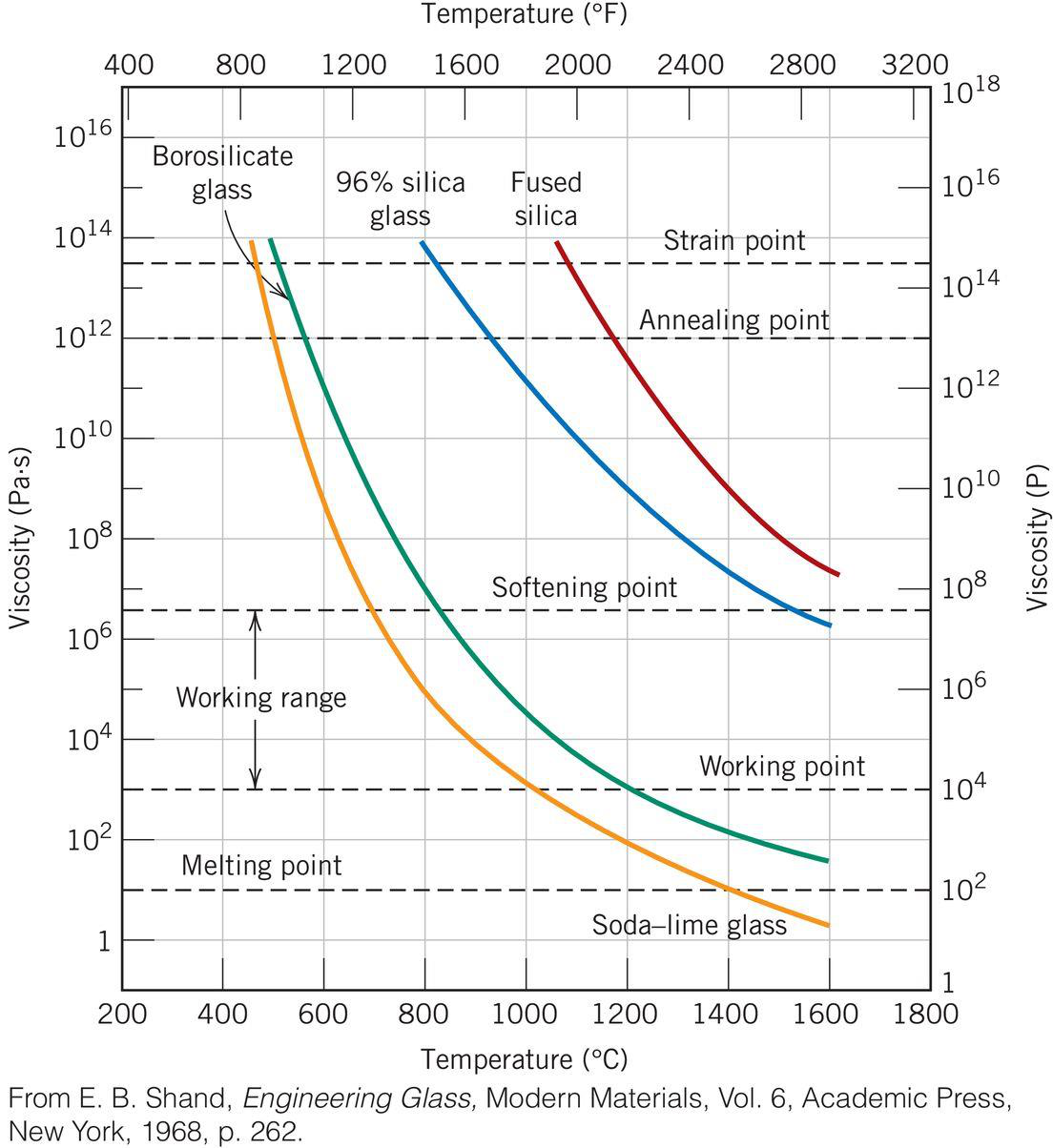
GLASS FORMING - VISCOSITY/TEMP
Key Points:
- Melting Point: Viscosity = 10 Pa-s
- Working Point: Viscosity = 1000 Pa-s
- Softening Point: Viscosity = 4 x 10^6 Pa-s
- Annealing Point: Viscosity = 10^12 Pa-s
- Strain Point: Viscosity = 3 x 10^3 Pa-s
Applications of Glass
GLASS IN THE CONSTRUCTION AND AUTOMOTIVE INDUSTRIES
- Integral part of modern buildings and vehicles, providing aesthetic and functional benefits.


Types of Glass
COMPOSITION OF GLASSES
Common Types:
- Soda-lime Glass (90%): Base composition includes Silica, Sodium, Calcium.
- Borosilicate Glass: Lower expansion, used in lab equipment.
- Fused Silica: High thermal resistance.
- Lead Glasses: High refractive index; used for optical applications.
- Gorilla Glass: Toughened for smartphone screens.
Advanced Applications
ADVANCED APPLICATIONS OF GLASS
- Self-cleaning, solar, and smart glasses with control features.

Conclusion
CONCLUSIONS
- Glasses play vital roles across various industries due to their properties and compositions.