Sign up to unlock more features
- Save this deck to your account
- Study flashcards with spaced repetition
- Export to Anki (.apkg) or PDF
- Process documents up to 100 pages
- Images extracted from PDFs and documents
- Better text extraction from your PDFs and documents
- Better flashcards with our more advanced AI model
Flashcards in this deck (195)
-
What fraction of human energy needs is provided by dietary triacylglycerols?
About one-third of energy needs
nutrition metabolism -
What percentage of energy needs of the mammalian heart and liver is met by fatty acid oxidation?
About 80%
metabolism bioenergetics -
Which energy source do many hibernating animals, such as grizzly bears, rely on almost exclusively?
Fats (fatty acids/triacylglycerols)
physiology hibernation -
For what type of energy needs are glucose and glycogen primarily used?
Short-term energy needs and quick delivery
metabolism carbohydrates -
For what type of energy needs are fats primarily used?
Long-term (months) energy storage with slow delivery
metabolism lipids -
Why do fatty acids carry more energy per carbon than carbohydrates?
Because fatty acids are more reduced, giving more energy per carbon
bioenergetics chemistry -
Why are fats advantageous for storage compared with carbohydrates regarding water weight?
Fats are nonpolar and therefore carry less associated water
physiology lipids -
bile liver gallbladder
-
What is the primary action of bile salts on dietary fats in the small intestine?
Bile salts emulsify dietary fats, forming mixed micelles in the small intestine.
bile emulsification digestion -
What is the role of intestinal lipases in fat digestion?
Intestinal lipases degrade triacylglycerols.
lipase digestion -
After degradation by lipases, what happens to fatty acids and breakdown products in the intestine?
They are taken up by the intestinal mucosa and converted into triacylglycerols.
absorption mucosa -
How are triacylglycerols transported from intestinal mucosa to tissues?
Triacylglycerols are incorporated with cholesterol and apolipoproteins into chylomicrons, which move through the lymphatic system and bloodstream to tissues.
chylomicron transport -
Which protein activates lipoprotein lipase in the capillary?
ApoC-II activates lipoprotein lipase in the capillary.
apoc-ii lipoprotein -
What reaction does lipoprotein lipase catalyze at the capillary?
Lipoprotein lipase converts triacylglycerols to fatty acids and glycerol.
lipoprotein lipase -
Once fatty acids enter myocytes or adipocytes, what are their two fates described?
- Oxidized as fuel (to CO₂ and ATP)
- Reesterified for storage
metabolism storage -
Name two bile acids that have an R4 group of '-NHCH₂COO' or '-NHCH₂CH₂SO₃' as listed.
- Glycocholic acid: R₄ = '-NHCH₂COO'
- Taurocholic acid: R₄ = '-NHCH₂CH₂SO₃'
bile chemistry -
What structural feature of bile acids is highlighted in the lipid–bile acid diagram?
Bile acids have a hydrophobic side and a hydrophilic side that interact with lipids to form micelles.
structure micelle -
Provide a concise summary of the sequence from dietary fat ingestion to fatty acid uptake by tissues.
1) Bile salts emulsify fats → 2) Intestinal lipases degrade triacylglycerols → 3) Intestinal mucosa converts products to triacylglycerols → 4) Chylomicrons transport them → 5) Lipoprotein lipase releases fatty acids for tissue uptake.
pathway overview -
Which image illustrates the overall process of fatty acid absorption including bile salt emulsification and chylomicron transport?
See diagram:
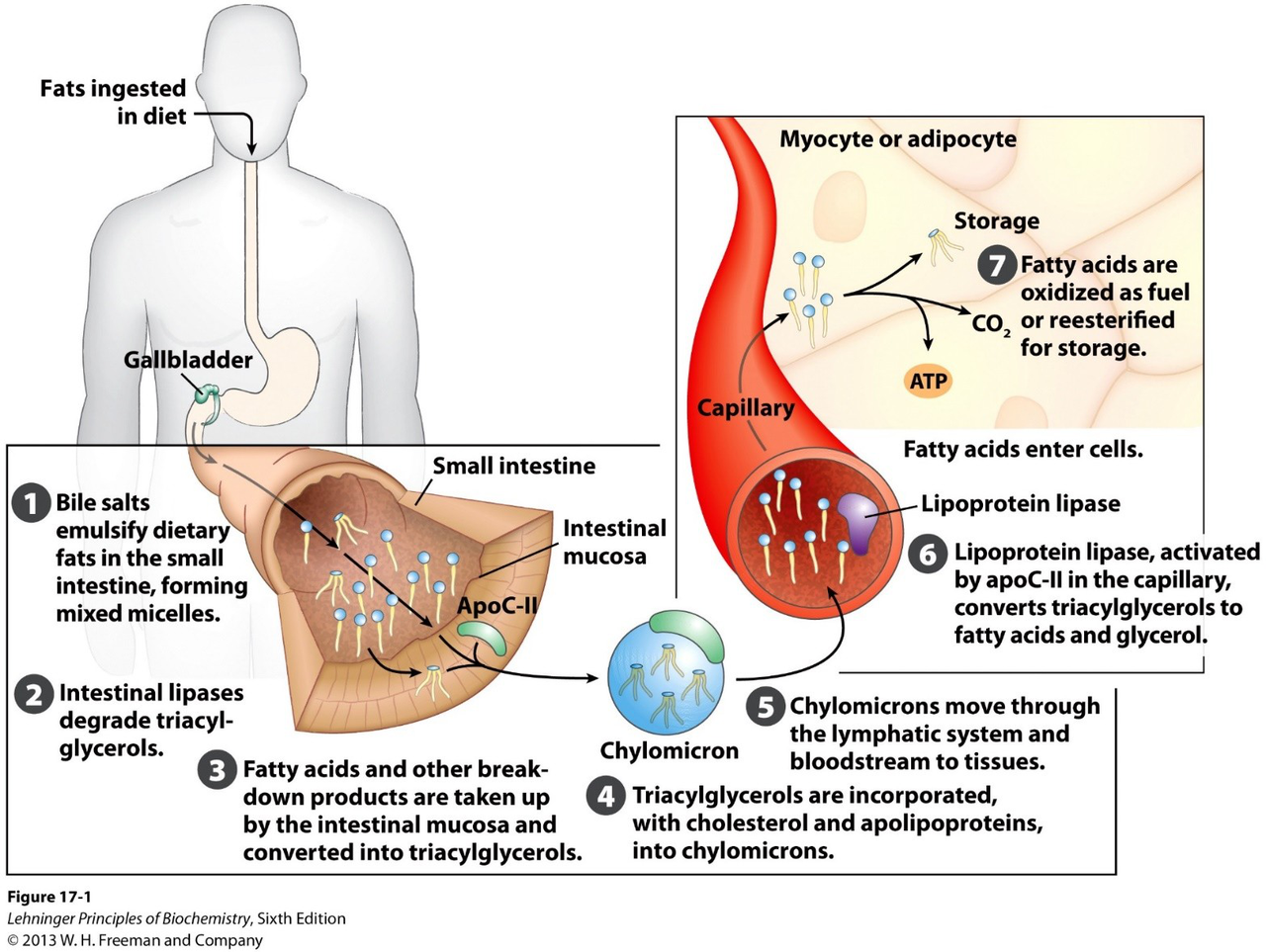 diagram visual
diagram visual -
How are dietary lipids transported in the blood to storage sites?
They are transported in chylomicrons and brought to adipocytes.
lipid transport chylomicron -
After release from adipocytes, what protein binds fatty acids for delivery to other tissues?
Albumin binds released fatty acids for delivery to other tissues.
fattyacids albumin transport -
apolipoprotein chylomicron
-
What property of apolipoproteins explains their role in lipid particles?
Apolipoproteins are amphipathic (like a detergent).
apolipoprotein structure -
Name the main lipid components present in chylomicrons.
- Triacylglycerols and cholesteryl esters
- Cholesterol
- Phospholipids
- Fatty acids
chylomicron lipids components -
Where is white fat primarily stored in the body?
- Subcutaneous connective tissue
- Abdominal (visceral) cavity
fat adipose -
beige adipose
-
What are the origins and primary function-related location of brown fat?
- Derived from muscle cells
- Found in shoulder blades, neck, along spinal cord, above collarbone, and around vital organs
brown adipose -
What signaling materials do adipocytes release to communicate with other organs?
- Adipokines (proteins, lipids, metabolites)
- Noncoding RNAs
- Extracellular vesicles
adipocytes signaling -
adipokines proteins
-
List three lipid or small-molecule adipose-derived factors mentioned.
- FAHFAs
- LPAs sphingolipids
- Uric acid uridine
lipids metabolites -
Which types of non-coding RNAs are specified as released by adipose tissue?
- lncRNAs
- miRNAs
- small RNAs
ncrna adipose -
What are the main cellular components shown for white, beige, and brown adipocytes?
- Lipid droplet
- Nucleus
- Mitochondrion
cell adipocyte -
lipolysis insulin metabolism
-
Which hormone activates adipocyte lipolysis during low glucose?
Glucagon activates lipolysis during low glucose.
lipolysis glucagon metabolism -
What second messenger rises after glucagon receptor activation to promote lipolysis?
cAMP levels increase after glucagon receptor activation.
camp signaling lipolysis -
Which kinase is activated by cAMP to phosphorylate lipolytic proteins?
Protein kinase A (PKA) is activated by cAMP.
pka signaling lipolysis -
Name two enzymes or proteins phosphorylated by PKA to promote triacylglycerol breakdown.
- Hormone-sensitive lipase (HSL)
- Perilipin
hsl perilipin lipolysis -
What are the immediate lipid products of triacylglycerol hydrolysis in adipocytes?
- Diacylglycerol
- Monoacylglycerol
- Free fatty acids (FFAs)
triacylglycerol lipolysis lipids -
How are released free fatty acids transported in the bloodstream?
Free fatty acids bind to serum albumin for transport in the bloodstream.
ffa albumin transport -
After uptake by muscle cells, into which pathways are fatty acids metabolized for energy?
- β-oxidation
- Citric acid cycle
- Respiratory chain (oxidative phosphorylation)
betaoxidation cac oxidativephosphorylation -
atp co2 oxidation
-
Which adipocyte enzyme abbreviations appear in the lipolysis diagram as initiating triglyceride breakdown?
- ATGL
- HSL
- MGL
atgl hsl mgl -
What reaction do lipases catalyze?
Lipases catalyze triacylglycerol hydrolysis into free fatty acids and glycerol.
lipid lipase -
Where does triacylglycerol hydrolysis occur in adipocytes?
Hydrolysis occurs in the cytoplasm of adipocytes.
adipocyte location -
How does epinephrine (adrenaline) affect metabolism according to the text?
Epinephrine responds to stress and signals 'we need energy now,' increasing gluconeogenesis.
epinephrine hormone -
How does glucagon affect metabolism according to the text?
Glucagon responds to low blood sugar and signals 'we are out of glucose,' increasing glycogenolysis.
glucagon hormone -
What is the combined effect of epinephrine and glucagon on triacylglycerol?
Both epinephrine and glucagon activate triacylglycerol breakdown.
regulation lipolysis -
What role does adenylate cyclase play in the hormone signaling diagram?
Adenylate cyclase converts ATP to cAMP (ATP → cAMP + PPi) in the signaling pathway.
signal camp -
How does insulin modulate the effect of glucagon/epinephrine on lipolysis?
Insulin stimulates a phosphatase that reverses the effect of glucagon/epinephrine.
insulin regulation -
How is hormone-sensitive lipase regulated in the pathway shown?
Protein kinase activation (via the signaling cascade) activates hormone-sensitive lipase; phosphatase inactivates it.
hsl regulation -
Do different lipases act on partially hydrolyzed glycerides?
Yes, different lipases work on diacylglycerol and monoacylglycerol.
lipase substrate -
Give an example of a mixed triacylglycerol mentioned in the text.
- 1-Stearoyl, 2-linoleoyl, 3-palmitoyl glycerol
triacylglycerol example -
How does dietary glycerol first enter the glycolytic pathway?
Glycerol is phosphorylated by glycerol kinase to form glycerol 3-phosphate using ATP.
metabolism glycerol glycolysis -
Which enzyme converts glycerol 3-phosphate to dihydroxyacetone phosphate and what cofactor is involved?
Glycerol 3-phosphate dehydrogenase converts glycerol 3-phosphate to dihydroxyacetone phosphate using NAD+ → NADH + H+.
enzymes redox glycerol -
What enzyme interconverts dihydroxyacetone phosphate and D-glyceraldehyde 3-phosphate?
Triose phosphate isomerase converts dihydroxyacetone phosphate to D-glyceraldehyde 3-phosphate.
enzymes glycolysis triose -
How many ATP are produced from glycerol catabolism through the described reactions (gross and net)?
The subsequent reactions produce 21 ATP (20 net).
bioenergetics atp glycerol -
What metabolic capabilities does glycerol entry into glycolysis enable?
It allows limited anaerobic catabolism of fats and enables gluconeogenesis, creating a potential futile cycle.
metabolism gluconeogenesis fat -
Pathway diagram: show the sequence from glycerol to glycolysis (use image for illustration).
- Pathway: Glycerol → (glycerol kinase) → glycerol 3-phosphate → (glycerol 3-phosphate dehydrogenase, NAD+→NADH+H+) → dihydroxyacetone phosphate → (triose phosphate isomerase) → D-glyceraldehyde 3-phosphate → Glycolysis
 pathway diagram glycerol
pathway diagram glycerol -
Where are fatty acids converted to fatty acyl-CoA inside the cell?
- Endoplasmic reticulum (ER)
- Outer mitochondrial membrane
fattyacid location metabolism -
What are three cellular fates that require conversion of fatty acids to fatty acyl-CoA?
- Transport
- β-oxidation
- Attachment to phospholipids
fattyacid function metabolism -
What is the first chemical step catalyzed by fatty acyl-CoA synthetase?
The carboxylate ion is adenylated by ATP to form a fatty acyl-adenylate and PPi.
mechanism enzyme activation -
How is fatty acyl-CoA formed from the enzyme-bound acyl-adenylate?
The thiol of coenzyme A attacks the acyl-adenylate, displacing AMP and forming the thioester fatty acyl-CoA.
mechanism coa activation -
What is the energetic change associated with the overall two-step activation of a fatty acid to fatty acyl-CoA?
- ΔG'° = -19 kJ/mol for the adenylation step
- ΔG'° = -15 kJ/mol for the two-step process
energetics thermodynamics activation -
Which molecule is hydrolyzed immediately after formation of fatty acyl-adenylate, and by what general process?
The pyrophosphate (PPi) released is immediately hydrolyzed to two Pi (inorganic phosphate).
pyrophosphate hydrolysis mechanism -
Use the diagram showing fatty acyl-CoA synthetase to review the activation steps (image on answer).
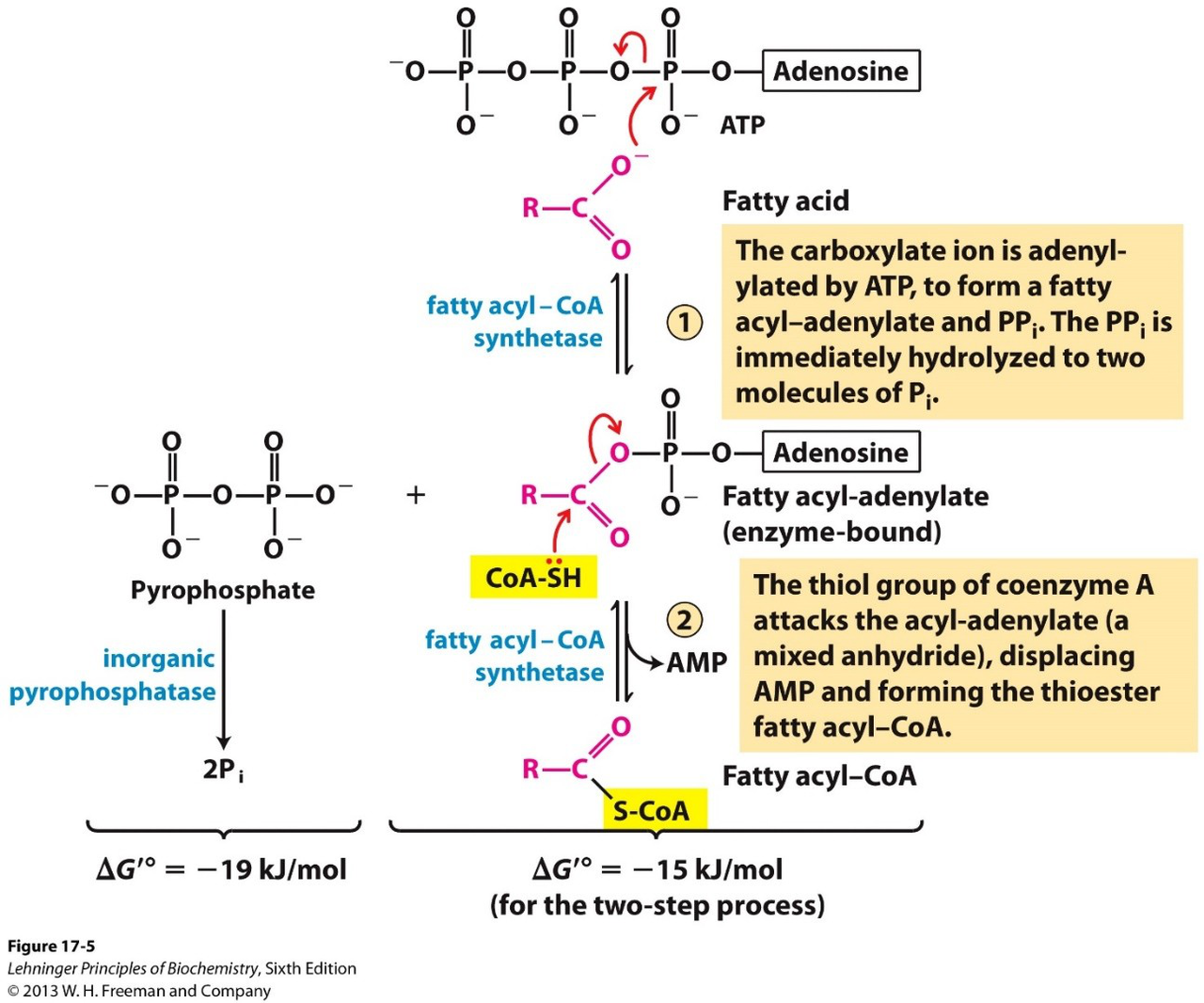 - ATP adenylates the fatty acid → acyl-adenylate + PPi
- CoA-SH attacks to give fatty acyl-CoA + AMPdiagram mechanism visual
- ATP adenylates the fatty acid → acyl-adenylate + PPi
- CoA-SH attacks to give fatty acyl-CoA + AMPdiagram mechanism visual -
biochemistry lipids transport
-
What enzyme attaches larger fatty acyl-CoAs to carnitine and where does this occur?
- Carnitine acyltransferase I (CAT I) in the outer mitochondrial membrane
biochemistry carnitine mitochondria -
How does the carnitine acyl-carnitine carrier move fatty acyl groups into the matrix?
- It swaps fatty acyl-carnitine for free carnitine, transporting the acyl-carnitine into the matrix and returning free carnitine to the intermembrane space
biochemistry transport carnitine -
What reaction does Carnitine acyltransferase II (CAT II) perform inside the matrix?
- Reverses CAT I to produce fatty acyl-CoA and carnitine
biochemistry carnitine beta-oxidation -
Where does β-oxidation of transported fatty acyl groups occur?
- In the mitochondrial matrix after fatty acyl-CoA is reformed by CAT II
biochemistry beta-oxidation mitochondria -
Illustrate the carnitine shuttle's main components and steps.
- Steps:
- CAT I attaches acyl to carnitine (outer membrane)
- Carrier swaps acyl-carnitine for carnitine across inner membrane
- CAT II reforms acyl-CoA in the matrix
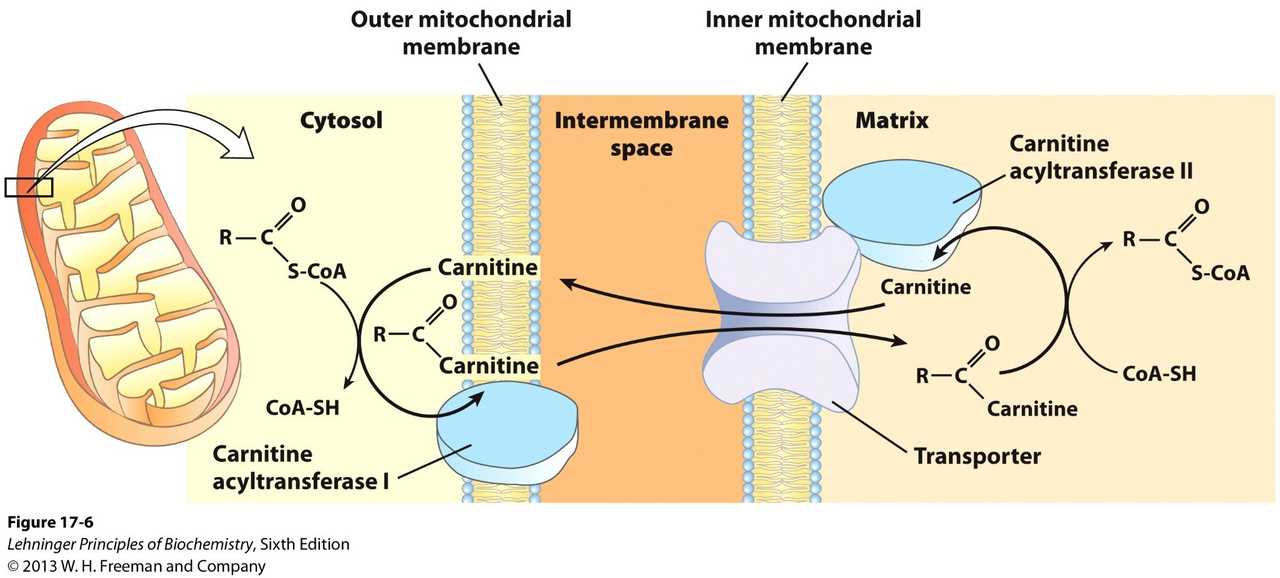 biochemistry carnitine diagram
biochemistry carnitine diagram -
metabolism fattyacid
-
What is the main outcome of Stage 1 (β-oxidation) of fatty acid oxidation?
Oxidative conversion of two-carbon units into acetyl-CoA, generating NADH and FADH2
β-oxidation energy -
What chemical transformation occurs to the fatty acyl-CoA β-carbon during β-oxidation?
Oxidation of the fatty acyl-CoA β-carbon to a thioester
β-oxidation chemistry -
What occurs in Stage 2 of fatty acid oxidation?
Oxidation of acetyl-CoA into CO₂ via the citric acid cycle, generating NADH and FADH2
citricacid metabolism -
What is produced in Stage 3 of fatty acid oxidation from NADH and FADH2?
ATP via oxidative phosphorylation (respiratory electron-transfer chain)
oxphos energy -
Name the main steps of the carnitine shuttle shown in the pathway.
- Acyl-CoA Synthetase
- CAT I
- Formation of Acylcarnitine (carnitine)
- Translocase
- CAT II
carnitine transport -
List the core enzymatic steps of the β-oxidation cycle shown in the diagram.
- Acyl-CoA Dehydrogenase
- Enoyl-CoA hydratase
- 3-hydroxyacyl-CoA dehydrogenase
- β-Ketothiolase
enzymes β-oxidation -
What are the immediate products after one cycle of β-oxidation according to the pathway text?
Shortened fatty acyl-CoA (−2C) plus one acetyl-CoA
products β-oxidation -
What are the electron carriers generated during stages 1 and 2 that feed into the respiratory chain?
NADH and FADH2
nadh fadh2 -
What are the final electron-transfer chain products listed in the diagram when ATP is produced?
- H₂O
- ATP (from ADP + Pi)
oxphos respiration -
What does each round of the β-oxidation cycle remove from the fatty acyl-CoA?
One 2‑carbon acetyl group removed as acetyl‑CoA.
biochemistry beta-oxidation -
How many β-oxidation rounds are required to oxidize palmitoyl‑CoA (C16)?
Seven rounds of β‑oxidation.
biochemistry palmitate -
How many acetyl‑CoA molecules are produced from one 16‑carbon palmitate?
- 8 acetyl‑CoA (8 × 2‑carbon units).
biochemistry energy -
What is the first enzymatic step in each β‑oxidation cycle and its cofactor change?
Acyl‑CoA dehydrogenase oxidizes the acyl‑CoA; FAD is reduced to FADH2.
enzymes beta-oxidation -
What reaction does enoyl‑CoA hydratase perform in β‑oxidation?
Enoyl‑CoA hydratase adds H2O across the double bond to form L‑β‑hydroxyacyl‑CoA.
enzymes hydration -
What is the role of β‑hydroxyacyl‑CoA dehydrogenase in the cycle and which cofactor is involved?
It oxidizes L‑β‑hydroxyacyl‑CoA to β‑ketoacyl‑CoA using NAD+, producing NADH + H+.
enzymes dehydrogenase -
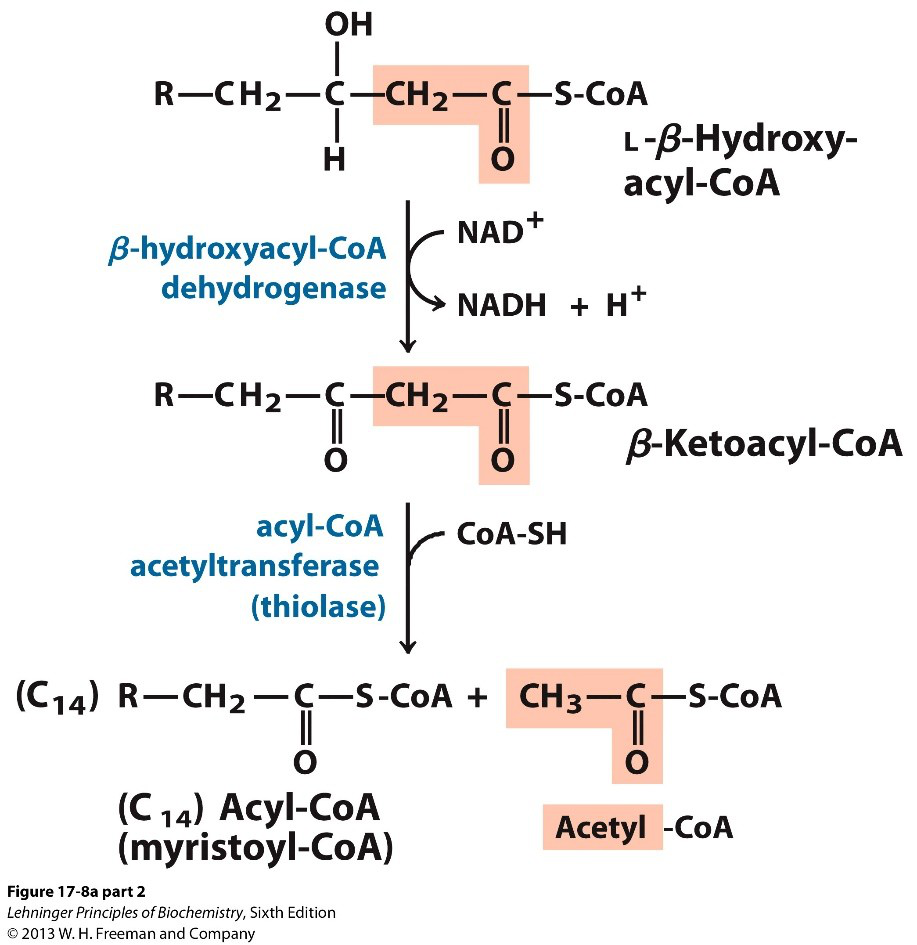
What reaction does acyl‑CoA acetyltransferase (thiolase) catalyze in β‑oxidation?
Thiolase cleaves β‑ketoacyl‑CoA to yield acetyl‑CoA and a shortened acyl‑CoA (e.g., C16 → C14 + acetyl‑CoA).
enzymes thiolase -
What is an example of the shortened acyl‑CoA after one round of palmitoyl‑CoA β‑oxidation?
Palmitoyl‑CoA (C16) becomes myristoyl‑CoA (C14) plus acetyl‑CoA.
examples palmitate -
List the main enzymes of one β‑oxidation cycle in order (compact summary).
- Acyl‑CoA dehydrogenase (FAD → FADH2)
- Enoyl‑CoA hydratase (adds H2O)
- β‑hydroxyacyl‑CoA dehydrogenase (NAD+ → NADH + H+)
- Acyl‑CoA acetyltransferase (thiolase)
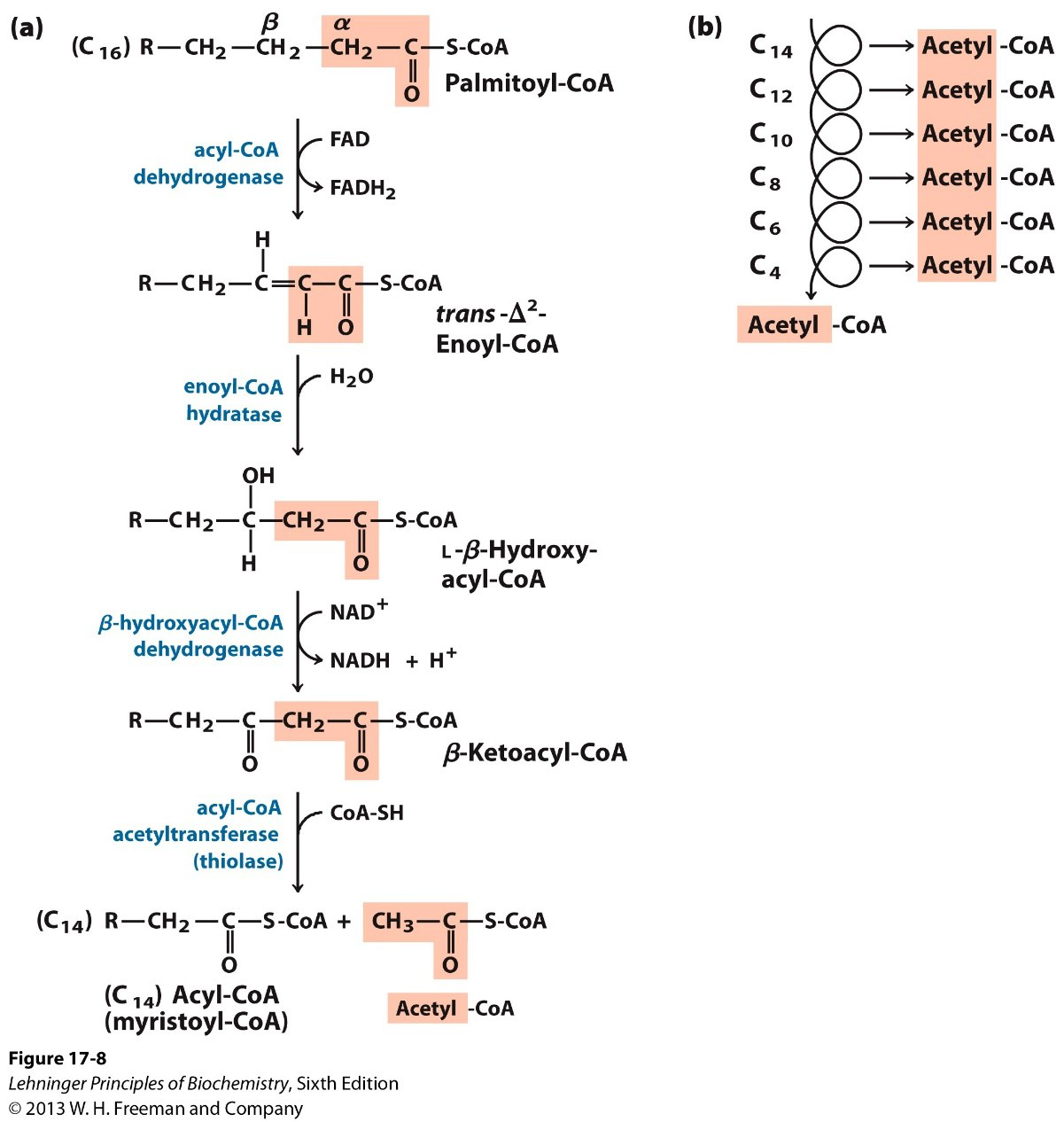 summary beta-oxidation enzymes
summary beta-oxidation enzymes -
Where do acyl-CoA dehydrogenases dehydrogenate alkanes to alkenes?
On the inner mitochondrial membrane
bioenergetics beta-oxidation -
What is the product stereochemistry formed by acyl-CoA dehydrogenase dehydrogenation?
A trans double bond
enzymes fattyacids -
Name the three chain-length specific acyl-CoA dehydrogenases listed.
- Very-long-chain AD (12-18 carbons)
- Medium-chain AD (4-14 carbons)
- Short-chain AD (4-8 carbons)
enzymes fattyacids -
cofactors beta-oxidation
-
How are electrons from the bound FAD of acyl-CoA dehydrogenase transferred to the electron transport chain?
They are transferred directly to the electron transport chain via an electron-transferring flavoprotein (ETF)
electrontransport etf -
Which citric acid cycle enzyme reaction is the acyl-CoA dehydrogenase step similar to?
The succinate dehydrogenase reaction
metabolism comparisons -
In β-oxidation, what chemical change occurs during the hydration (Step 2)?
The double bond (alkene) is hydrated with water to produce an alcohol catalyzed by enoyl-CoA hydratases.
biochemistry beta-oxidation -
Which enzymes catalyze the hydration of enoyl-CoA?
- Soluble short-chain hydratase (crotonase)
- Membrane-bound long-chain hydratase (part of the trifunctional complex)
enzymes hydratase -
What is the stereospecific product of enoyl-CoA hydration?
L-β-hydroxy-acyl-CoA (L-B-hydroxy-acyl-CoA).
 products stereochemistry
products stereochemistry -
How does the enoyl-CoA hydration compare to a citric acid cycle reaction?
It is similar to the fumarase reaction in the citric acid cycle and has the same stereospecificity.
comparison citricacidcycle -
Which enzyme catalyzes the dehydrogenation of L-β-hydroxyacyl-CoA to β-ketoacyl-CoA?
- β-hydroxyacyl-CoA dehydrogenase
 biochemistry beta-oxidation enzymes
biochemistry beta-oxidation enzymes -
What cofactor acts as the hydride acceptor for β-hydroxyacyl-CoA dehydrogenase?
- NAD+ (reduced to NADH + H+)
biochemistry cofactors redox -
The dehydrogenation by β-hydroxyacyl-CoA dehydrogenase is similar to which citric acid cycle reaction?
- Malate dehydrogenase reaction
biochemistry citricacidcycle comparisons -
What is the overall net chemical event in the chain transfer step of β-oxidation?
Thiolysis of a carbon–carbon bond, yielding a shortened acyl‑CoA and acetyl‑CoA.
biochemistry beta-oxidation mechanism -
Which carbon in 3-ketoacyl‑CoA is electrophilic during chain transfer?
The carbonyl carbon of 3-ketoacyl‑CoA is electrophilic.
biochemistry reactivity -
What role does the enzyme active site thiolate play in the chain transfer step?
The active site thiolate acts as a nucleophile that attacks and releases acetyl‑CoA from the substrate.
enzymology biochemistry -
How does CoA‑SH participate in transferring the fatty acid chain during thiolysis?
The terminal sulfur of CoA‑SH acts as a nucleophile and accepts the fatty acid chain from the enzyme to form a new acyl‑CoA.
biochemistry cofactors -
What are the two products formed after thiolysis in the chain transfer step?
- Shortened acyl‑CoA (2 carbons shorter)
- Acetyl‑CoA
products beta-oxidation -
Give a brief description of the chain transfer step illustrated in the diagram.
3‑Ketoacyl‑CoA undergoes enzyme‑mediated thiolysis: enzyme thiolate forms intermediate releasing acetyl‑CoA; CoA‑SH attacks to produce a shortened acyl‑CoA.
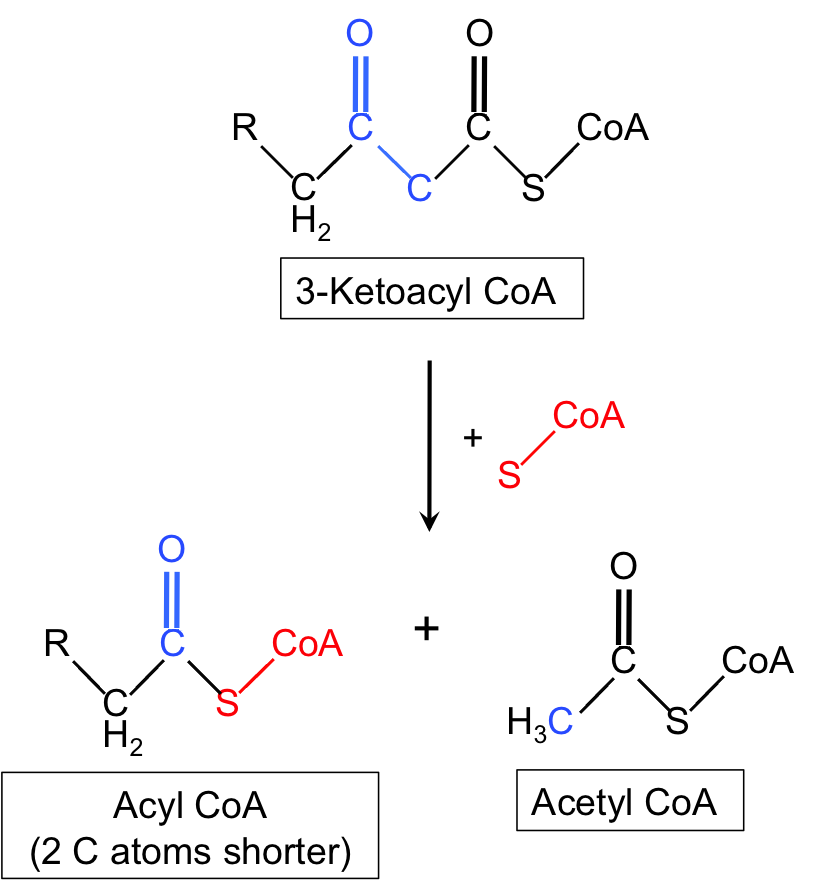 diagram visual
diagram visual -
Which enzyme complex catalyzes steps 2–4 of fatty acid β‑oxidation for chains ≥12 carbons?
The trifunctional protein (TFP) complex catalyzes steps 2–4 and processes fatty acid chains with 12 or more carbons.
β-oxidation tfp metabolism -
What is the quaternary structure of the trifunctional protein and one functional consequence?
TFP is a hetero-octamer, and this quaternary structure allows substrate channeling.
tfp structure substrate_channeling -
Which enzymatic activities are carried on the four TFP alpha (a) subunits?
- 2,3-Enoyl-CoA hydratase
- 3-Hydroxyacyl-CoA dehydrogenase
- Inner mitochondrial membrane binding
tfp alpha enzymes -
What activity do the four TFP beta (ẞ) subunits provide?
The four TFP ẞ subunits provide long-chain 3-ketoacyl-CoA thiolase activity.
tfp beta thiolase -
How are shorter fatty acyl chains (less than 12 carbons) processed after TFP action?
Soluble enzymes in the mitochondrial matrix process shorter chains, including those broken down by TFP to less than 12 carbons.
β-oxidation matrix short-chain -
How does TFP function change with age and what molecule was shown to affect FA oxidation and CD8+ T cells?
TFP function decreases with age; spermidine, a natural polyamine, increased fatty acid oxidation and rejuvenated CD8+ T cells.
aging spermidine tfp -
What does the provided diagram illustrate about TFP and related components?
Illustration of the TFP complex, acyl-CoA dehydrogenases, TFPα (hydratase and dehydrogenase), TFPẞ (thiolase), MOM, MIM, OXPHOS, and the TCA cycle
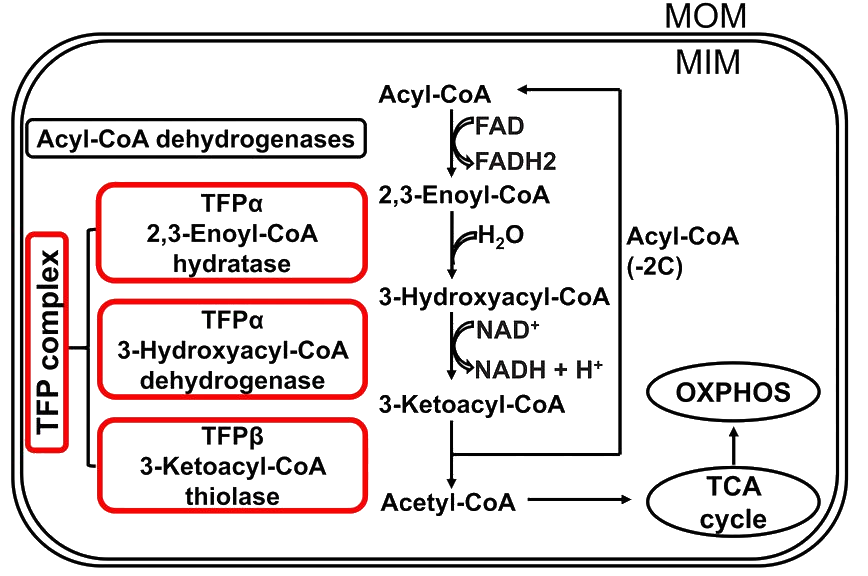 diagram tfp visual
diagram tfp visual -
Write the overall reaction for one cycle of fatty acid β-oxidation (substrate C-acyl-CoA).
C-acyl-CoA + FAD + NAD+ + H₂O + COA → Cn-2-acyl-CoA + FADH2 + NADH + H+ + acetyl-CoA
beta-oxidation reaction -
palmitate cycles
-
palmitate acetyl-coa
-
What are the NADH, FADH2 yields per palmitate from the β-oxidation dehydrogenase steps listed?
- 7 FADH2 (acyl-CoA dehydrogenase)
- 7 NADH (β-hydroxyacyl-CoA dehydrogenase)
palmitate reducing-equivalents -
What is the ATP yield assumed per NADH and per FADH2 during oxidative phosphorylation in these calculations?
- 2.5 ATP per NADH oxidized
- 1.5 ATP per FADH2 oxidized
oxidative-phosphorylation atp-yield -
What is the total ATP yield from complete oxidation of one palmitoyl-CoA to CO₂ and H₂O (per the table)?
108 ATP
palmitoyl-coa atp -
Per acetyl-CoA entering the citric acid cycle, what reduced cofactors and high-energy product are formed?
- 3 NADH
- 1 FADH2
- 1 GTP
citric-acid-cycle products -
Show the table image illustrating ATP yield from palmitoyl-CoA oxidation (use as study aid).
- Table: Yield of ATP during Oxidation of One Molecule of Palmitoyl-CoA to CO₂ and H₂O

table visual -
Show the diagram image of fatty acid catabolism (use as study aid).
- Diagram: fatty acid catabolism and acetyl-CoA production
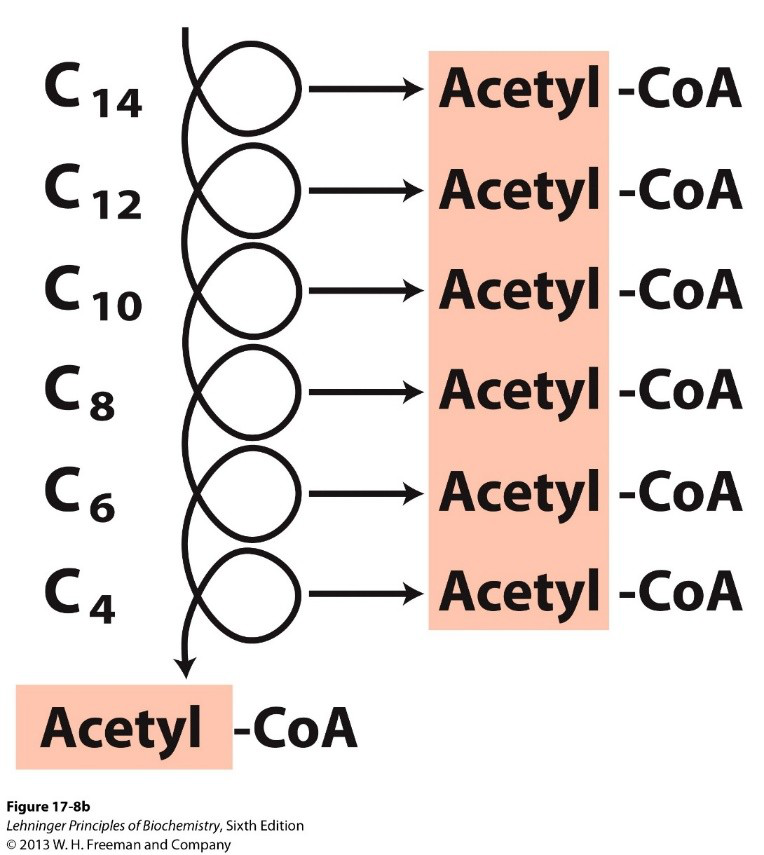
diagram visual -
What general observation is made about cellular processes?
Cellular processes often have similar pathways.
cellular pathways -
What is β-oxidation described as in the text?
A conserved reaction sequence to convert a β-carbon to a carbonyl.
beta-oxidation metabolism -
Name the three processes compared in the provided diagram.
- β Oxidation
- Citric acid cycle
- Oxidation of isoleucine (leucine, valine)
pathways comparison -
What type of double bonds do unsaturated fatty acids contain, and are they substrates for enoyl-CoA hydratase (Step 2)?
- Contain: cis double bonds
- Substrate for enoyl-CoA hydratase (Step 2)? No
beta-oxidation unsaturated lipid -
What enzymatic step is required for monounsaturated fatty acid β-oxidation when a cis double bond starts at carbon 3?
An isomerase converts the cis double bond at carbon 3 to a trans double bond (Δ3 → Δ2 isomerization).
lipid isomerase unsaturated -
In the provided Oleoyl-CoA pathway, what intermediate is formed after three β-oxidation cycles and which enzyme converts it to a trans-Δ2 form?
- Intermediate: cis-Δ3-Dodecenoyl-CoA
- Enzyme: Δ3, Δ2-enoyl-CoA isomerase converts it to trans-Δ2-Dodecenoyl-CoA
oleoyl isomerase pathway -
According to the diagram transcript, how many Acetyl-CoA units are produced after the initial three β-oxidation cycles of Oleoyl-CoA, and how many after the subsequent five cycles?
- After three cycles: 3 Acetyl-CoA
- After five cycles: 6 Acetyl-CoA
acetyl-coa beta-oxidation oleoyl -
Where should the diagram illustrating Δ3, Δ2-enoyl-CoA isomerase be placed for a question about the image contents?
Use the diagram on the question side when asking about structures or processes visible in the image:
 image diagram media
image diagram media -
Which enzymes are required for polyunsaturated fatty acid β-oxidation?
- An enoyl-CoA isomerase
- A 2,4-dienoyl-CoA reductase (reductase that reduces cis double bonds not at carbon 3)
biochemistry lipids -
What does the first double bond encountered in polyunsaturated FA β-oxidation require?
Isomerization
biochemistry fattyacid -
What does the second double bond encountered in polyunsaturated FA β-oxidation require?
Reduction and/or isomerization
biochemistry fattyacid -
biochemistry substrate
-
How many acetyl-CoA molecules are produced after the three β-oxidation cycles shown for linoleoyl-CoA?
3 Acetyl-CoA
biochemistry energy -
Which two specific enzymes are highlighted in the diagram of polyunsaturated FA β-oxidation?
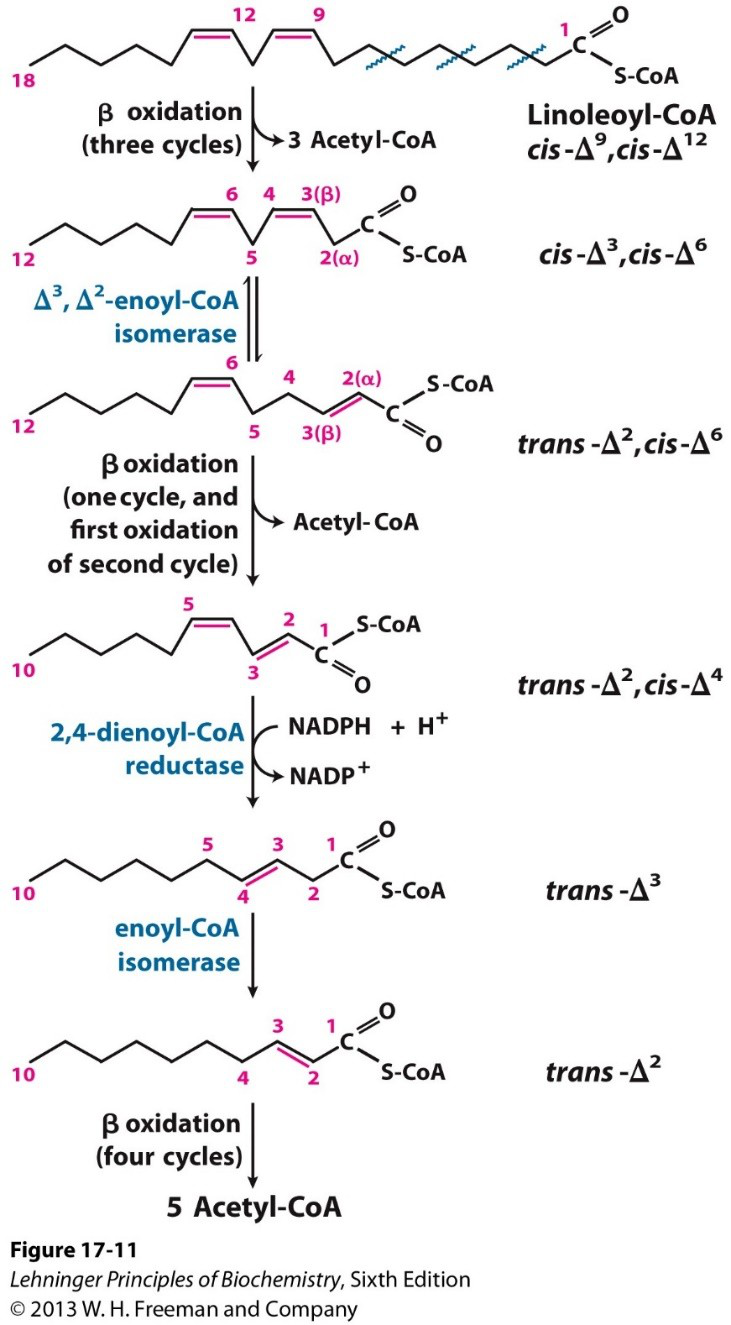
- Δ3, Δ2-enoyl-CoA isomerase
- 2,4-dienoyl-CoA reductase
biochemistry enzymes image -
fattyacids biosynthesis
-
betaoxidation metabolism
-
gluconeogenesis metabolism
-
Name two biological sources or routes that produce propionyl-CoA besides odd-chain β-oxidation.
- Bacterial metabolism in the rumen of ruminants
- Breakdown of cholesterol and several amino acids
rumen catabolism -
lipid metabolism cofactor
-
biochemistry cofactor traceelement
-
How is coenzyme B12 formed in a rare reaction involving ATP?
By a reaction that cleaves the triphosphate from ATP
biochemistry biosynthesis -
Which conversion in odd-chain fatty acid metabolism requires coenzyme B12?
- Conversion of L-methylmalonyl-CoA to succinyl-CoA
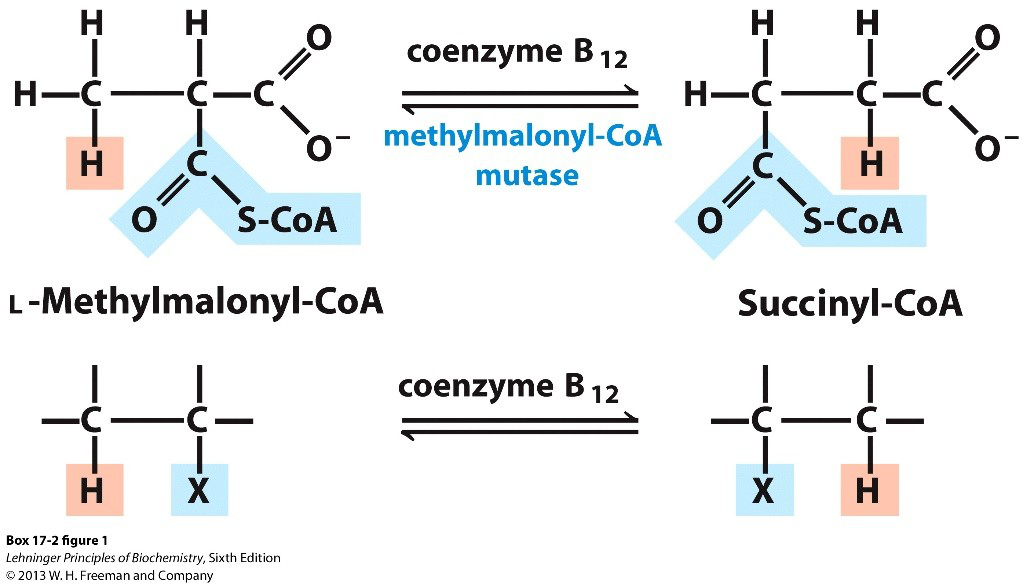 fattyacid metabolism methylmalonyl
fattyacid metabolism methylmalonyl -
hematology vitamin b12
-
What condition results from vitamin B12 deficiency due to intestinal malabsorption?
Pernicious anemia
pathology anemia b12deficiency -
Name two clinical consequences of vitamin B12 deficiency mentioned in the text.
- Reduced erythrocyte production and low hemoglobin
- CNS impairment
clinical b12 symptoms -
treatment b12 therapy
-
What structural features are highlighted in the coenzyme B12 molecular diagram?
- Corrin ring system and cobalt ion
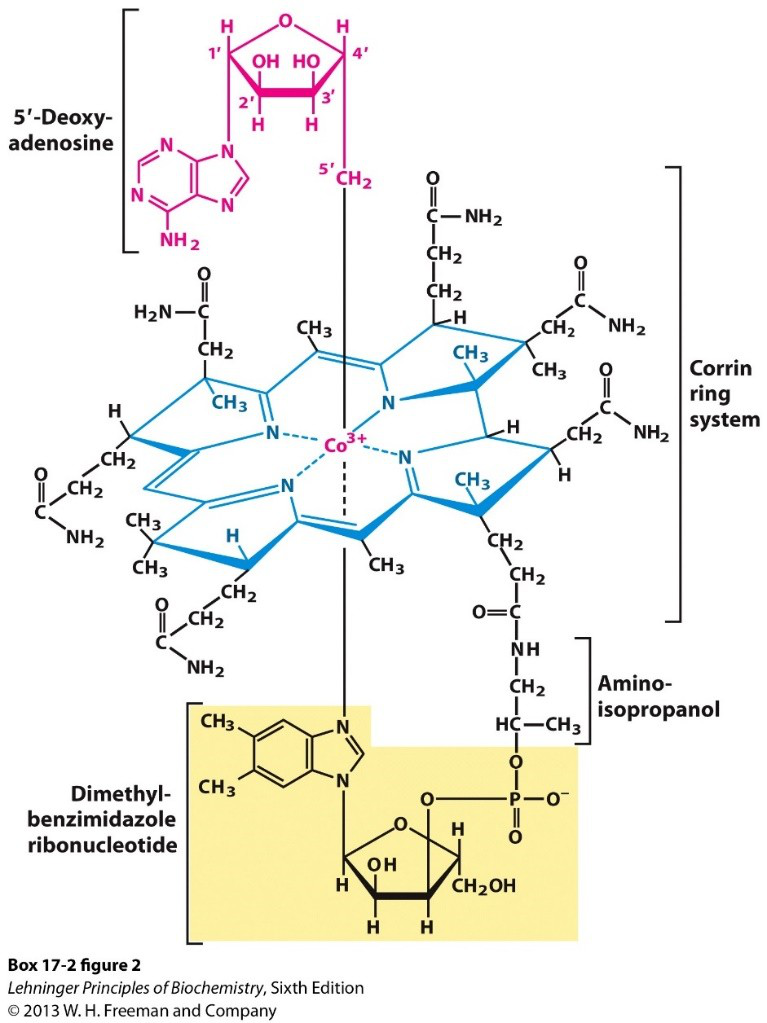 structure b12 corrin
structure b12 corrin -
What effect does insulin have on fatty acid metabolism?
Insulin activates fatty acid synthesis by causing acetyl-CoA carboxylase to be dephosphorylated.
metabolism lipids insulin -
What effect does glucagon have on fatty acid metabolism?
Glucagon activates fatty acid breakdown and causes acetyl-CoA carboxylase to be phosphorylated.
metabolism lipids glucagon -
Where does most β-oxidation occur in animals?
Mitochondria (with mitochondrial acyl-CoA dehydrogenase passing electrons into the respiratory chain)
biochemistry animals mitochondria -
What role do peroxisomes play in animal fatty acid β-oxidation?
They are used only to breakdown very long fatty acids (≥ 22 carbons).
biochemistry peroxisome animals -
How are electrons from mitochondrial acyl-CoA dehydrogenase transferred in animals and what is the energy outcome?
Electrons pass to an electron-transferring flavoprotein and into the respiratory chain; energy is captured as ATP.
biochemistry electrontransfer atp -
In plants, where does β-oxidation occur in leaves and in seeds?
Leaves: peroxisomal β-oxidation; Seeds: glyoxysomal β-oxidation.
plants peroxisome glyoxysome -
In plant peroxisomal/glyoxysomal β-oxidation, what is the final electron acceptor and what happens to the released energy?
Electrons are passed directly to molecular oxygen (O2) and the energy is released as heat.
plants oxidation energy -
Show a diagram comparing mitochondrial versus peroxisomal/glyoxysomal β-oxidation.

Use this diagram to review differences in electron transfer and energy outcome between mitochondrion and peroxisome/glyoxysome.
diagram comparison visual -
Show a diagram of peroxisome/glyoxysome pathways including VLCFA processing.
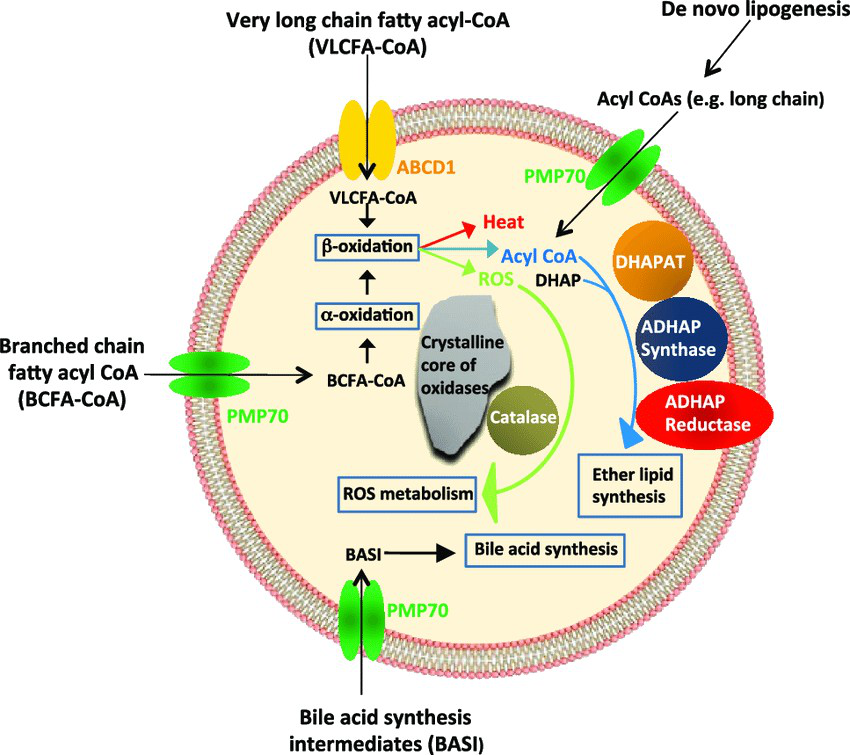
Refer to it to visualize VLCFA-CoA and BCFA-CoA processing, ROS metabolism, and bile acid synthesis links.
diagram peroxisome vlcfa -
Show a diagram of glyoxysome seed triacylglycerol conversion to glucose.

Use this to review conversion of seed triacylglycerols through β-oxidation and the glyoxylate cycle to gluconeogenesis and glucose.
diagram glyoxysome gluconeogenesis -
What are 'ketone bodies' in biochemical metabolism?
Water-soluble molecules produced from acetyl-CoA when glucose is low; formed by ketogenesis in the liver.
biochemistry ketone metabolism -
When does the body convert fats (as acetyl-CoA) into ketone bodies?
During times of low glucose the liver converts fats (acetyl-CoA) into ketone bodies via ketogenesis.
metabolism ketogenesis energy -
What is the first enzymatic step in ketogenesis described in the notes?
A thiolase reaction that joins two acetate units; it is the reverse of the last step in β-oxidation.
enzymes thiolase ketogenesis -
ketone compounds biochemistry
-
Show a diagram of the ketogenesis pathway that starts from 2 Acetyl-CoA.
Pathway diagram:
 (Thiolase → HMG-CoA synthase → HMG-CoA lyase → Acetoacetate / ẞ-hydroxybutyrate)diagram ketogenesis pathway
(Thiolase → HMG-CoA synthase → HMG-CoA lyase → Acetoacetate / ẞ-hydroxybutyrate)diagram ketogenesis pathway -
Provide an image of the molecular structure of acetoacetate.
Molecular structure:
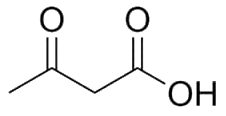 Name: Acetoacetatestructure acetoacetate ketone
Name: Acetoacetatestructure acetoacetate ketone -
Provide an image of the molecular structure of ẞ-hydroxybutyrate.
Molecular structure:
 Name: ẞ-hydroxybutyratestructure beta-hydroxybutyrate ketone
Name: ẞ-hydroxybutyratestructure beta-hydroxybutyrate ketone -
In which tissues does the provided diagram show ketone body production and related processes?
- Liver
- Skeletal muscle
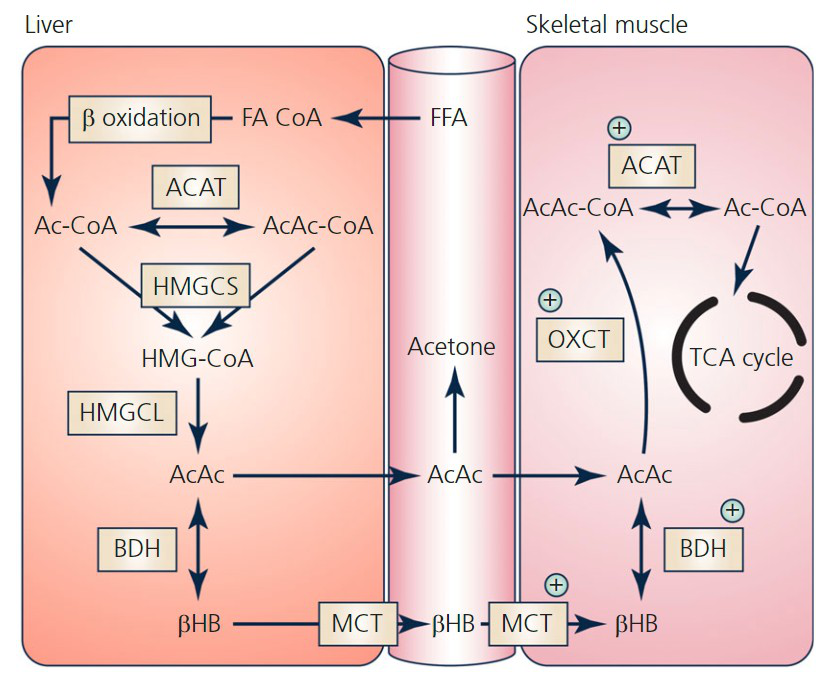 ketogenesis tissue
ketogenesis tissue -
Name three key metabolites labeled in the ketone body production diagram.
- Acetyl‑CoA (Ac‑CoA)
- Acetoacetate (AcAc)
- β‑Hydroxybutyrate (BHB)
 metabolites ketone
metabolites ketone -
enzymes ketogenesis
-
tca metabolism
-
Which physiological states are plotted on the plasma βHB concentration graph?
- Post‑prandial
- Fasted states
- Ketogenic diet
- Starvation
- Diabetic ketoacidosis
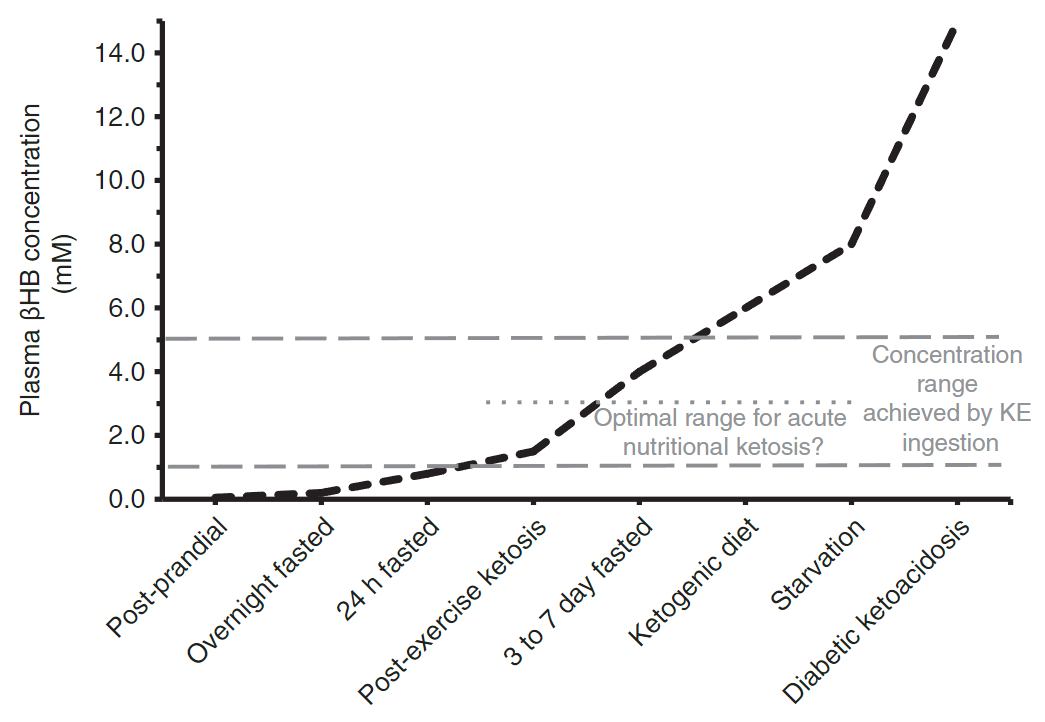 βhb physiology
βhb physiology -
What does the plasma βHB graph indicate about concentration ranges?
- It shows changing plasma βHB concentration (mM) across physiological states with indicated optimal concentration ranges.
 βhb concentration
βhb concentration -
Why is acetyl-CoA converted into ketone bodies when oxaloacetate is depleted?
Because entry of acetyl-CoA into the citric acid cycle requires oxaloacetate, so when oxaloacetate is depleted acetyl-CoA is diverted to ketone body formation.
ketone metabolism -
How does ketone body formation affect CoA availability for ẞ-oxidation?
Conversion of acetyl-CoA into ketone bodies frees CoA, allowing continued ẞ-oxidation.
beta-oxidation cofactor -
Where are ketone bodies synthesized and can the liver use them for its own energy?
Ketone bodies are made in the liver, but the liver lacks ẞ-ketoacyl-CoA transferase so it cannot use them for energy.
liver ketone -
How do ketone bodies reach other tissues?
Ketone bodies travel from the liver to other tissues through the bloodstream.
transport circulation -
What happens to ketone bodies once they enter a peripheral cell?
They are imported into the mitochondria and converted into acetyl-CoA, which then enters the citric acid cycle.
mitochondria ketone -
Can acetyl-CoA produced from ketone bodies be converted back into pyruvate or glucose?
No, acetyl-CoA cannot be converted back to pyruvate or glucose.
acetyl-coa gluconeogenesis -
Which enzymes are named in the pathway converting ketone bodies back to acetyl-CoA?
The pathway is shown as proceeding via ẞ-ketoacyl-CoA transferase and thiolase.
enzymes ketone -
What happens to ketone body production in healthy, well-nourished individuals?
Healthy, well-nourished individuals produce ketone bodies at a relatively low rate.
ketogenesis metabolism -
Under what metabolic conditions do ketone bodies increase and why?
During starvation and diabetes, cellular glucose is low, oxaloacetate is depleted, and acetyl-CoA accumulates, increasing ketone production.
ketogenesis diabetes starvation -
Where are ketone bodies synthesized and how are they distributed?
Ketone bodies are made in the liver mitochondrial matrix and released into the bloodstream.
liver mitochondria transport -
Which organs use ketone bodies as an energy source?
Ketone bodies are used by the brain, heart, and muscle.
energy organs -
What are the clinical consequences of high ketone body levels?
High ketone body levels lower blood pH, causing ketoacidosis.
pathology acidosis -
What causes the characteristic breath odor in ketosis or ketoacidosis?
High blood acetone results in a characteristic breath odor.
acetone symptom -
Name the two ketone bodies exported as energy sources mentioned in the diagram transcript.
- Acetoacetate
- D-β-hydroxybutyrate
ketones names -
Use the diagram of hepatic ketogenesis as an illustration of ketone export (image on answer).
Diagram:
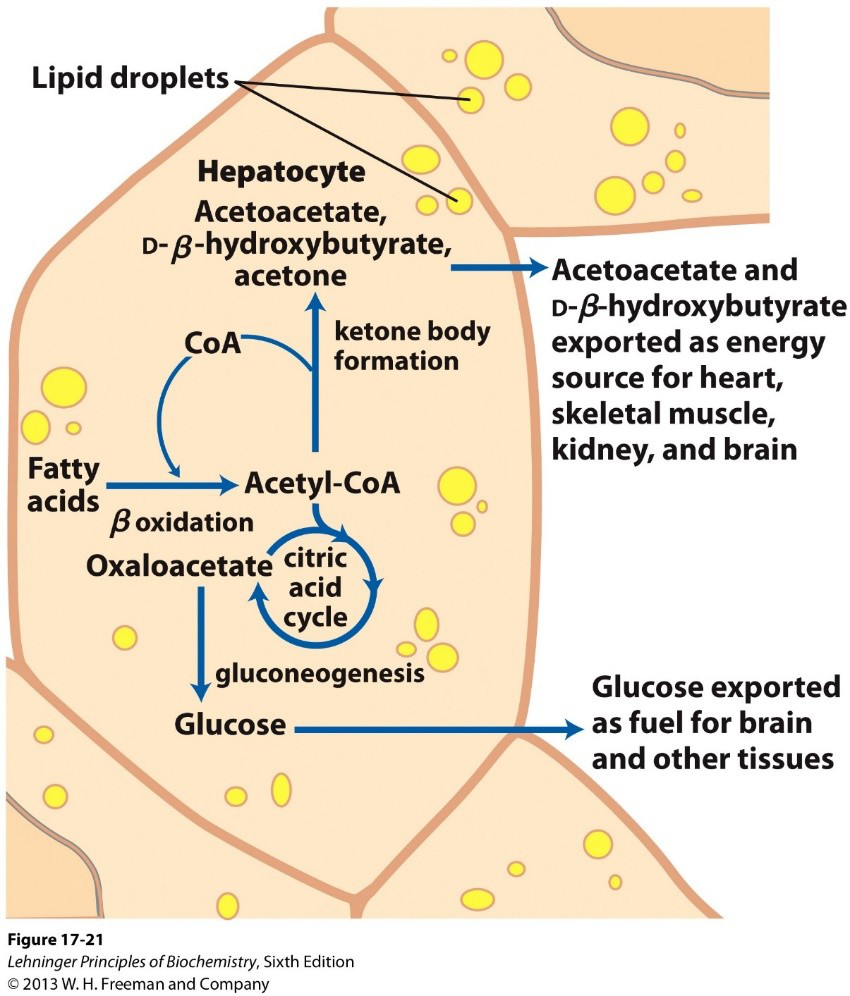 This shows acetoacetate and D-β-hydroxybutyrate exported as energy sources for heart, skeletal muscle, kidney, and brain.diagram illustration
This shows acetoacetate and D-β-hydroxybutyrate exported as energy sources for heart, skeletal muscle, kidney, and brain.diagram illustration -
Why can the adult brain not rely primarily on fatty acids for energy?
- Fatty acids are only minimally used for fuel because ẞ-oxidation requires a lot of oxygen and creates superoxides, risking neuronal hypoxia.
brain metabolism fattyacids -
brain energy ketones
-
Why is glucose preferred over fatty acids for ATP generation in neurons?
- Glucose provides faster ATP generation than fatty acids.
glucose atp neurons -
Give one reason albumin does not deliver fatty acids to the brain.
- Albumin doesn't cross the blood-brain barrier.
albumin bbb transport -
astrocytes ketogenesis brain
Overview: Why fats matter
- Fats are a major long-term energy store; ~one-third of dietary energy comes from triacylglycerols.
- Organs like heart and liver obtain ~80% of their energy from fatty acid oxidation.
- Fats store more energy per carbon (more reduced) and less water (nonpolar) compared with carbohydrates.
Fat digestion and absorption
- Emulsification: Bile salts (made in liver, stored in gallbladder) emulsify dietary fats into mixed micelles for digestion.
 Alt: Diagram of intestinal fat absorption and mixed micelle formation.
Alt: Diagram of intestinal fat absorption and mixed micelle formation. - Hydrolysis: Intestinal lipases degrade triacylglycerols to free fatty acids (FFAs) and monoacylglycerols.
- Uptake & reassembly: Enterocytes uptake breakdown products and re-form triacylglycerols.
- Chylomicron assembly: Triacylglycerols + cholesterol + apolipoproteins (e.g., ApoB-48, ApoC-II) form chylomicrons for lymph/blood transport.
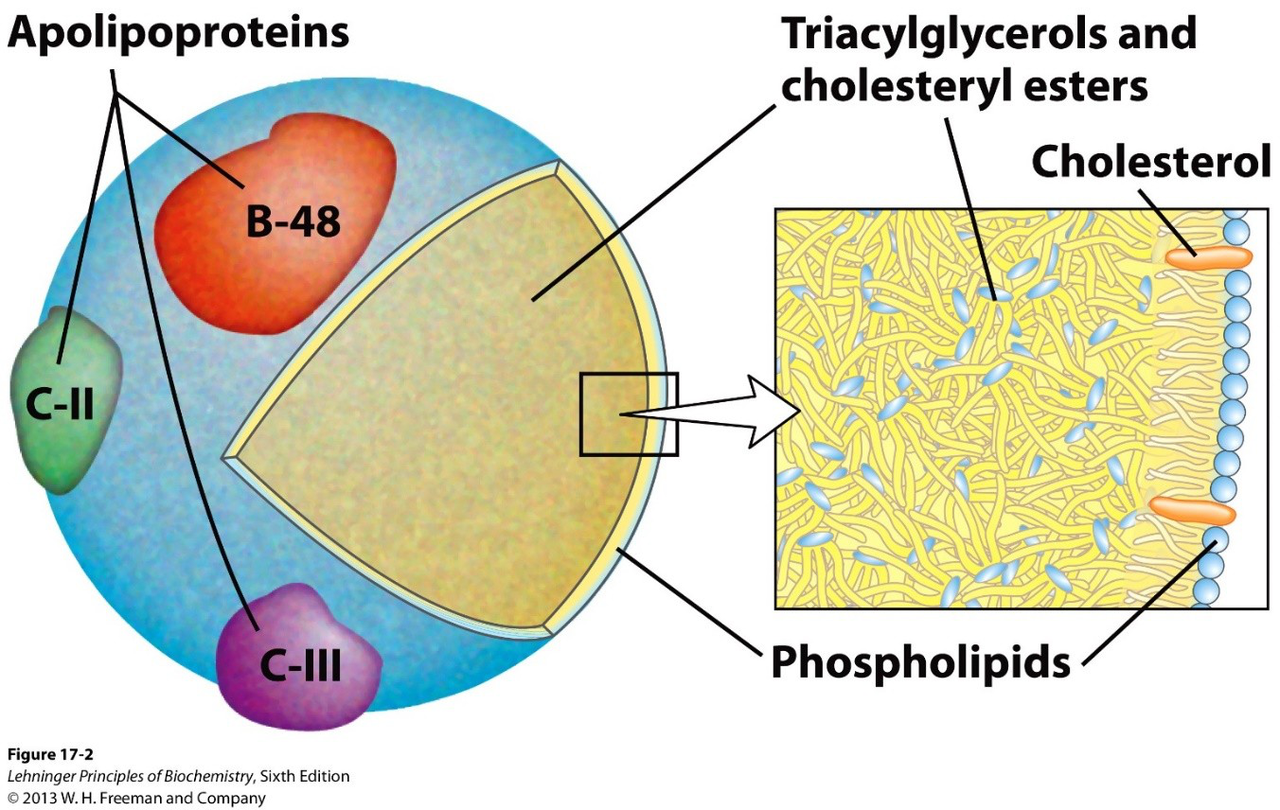 Alt: Chylomicron showing apolipoproteins and lipid cargo.
Alt: Chylomicron showing apolipoproteins and lipid cargo.
Lipid transport in blood
- Large lipids travel as lipoproteins (chylomicrons, VLDL, LDL, HDL).
- Released FFAs bind albumin for delivery to tissues that will oxidize them.
Fat storage: adipose tissue types and signaling
- White adipose: energy storage (subcutaneous, visceral).
- Beige adipose: white-derived, inducible thermogenic function (cold/exercise).
- Brown adipose: many mitochondria; specialized for heat production.
 Alt: Comparison of white, beige, and brown adipocytes.
Alt: Comparison of white, beige, and brown adipocytes. - Adipocytes secrete adipokines, lipids, noncoding RNAs and vesicles that signal to other organs.
Fat release (lipolysis) — regulation and mechanism
- Insulin inhibits lipolysis; glucagon and epinephrine activate it via cAMP → PKA.
- Key enzymes: ATGL, HSL (hormone-sensitive lipase), MGL; PKA phosphorylation activates HSL and perilipin remodeling.
 Alt: Lipolysis regulation flowchart.
Alt: Lipolysis regulation flowchart. - FFAs released enter blood bound to albumin or are taken up by nearby tissues for oxidation or re-esterification.
Triacylglycerol hydrolysis & glycerol fate
- Lipases hydrolyze triacylglycerol → diacylglycerol → monoacylglycerol → glycerol + FFAs.
- Glycerol is phosphorylated by glycerol kinase to glycerol-3-phosphate, oxidized to dihydroxyacetone phosphate, and enters glycolysis/gluconeogenesis.
 Alt: Glycerol to DHAP and glycolysis pathway.
Alt: Glycerol to DHAP and glycolysis pathway. - Net energy yield from glycerol catabolism is modest (stated as ~20 net ATP in original lecture context for activated routes).
Activation of fatty acids (formation of fatty acyl‑CoA)
- Fatty acids are converted to fatty acyl‑CoA by fatty acyl‑CoA synthetase (acyl‑CoA ligase) using ATP and CoA-SH.
- Reaction mechanism: adenylation (fatty acyl‑AMP) followed by CoA attack to form the thioester; PPi hydrolysis drives the reaction forward.
 Alt: Fatty acyl‑CoA synthetase reaction scheme.
Alt: Fatty acyl‑CoA synthetase reaction scheme.
Carnitine shuttle (transport into mitochondria)
- Short-chain FA (<C12) diffuse into mitochondria; long-chain FA require the carnitine shuttle.
- CAT I (outer membrane) converts acyl‑CoA → acyl‑carnitine; transporter moves it across inner membrane; CAT II regenerates acyl‑CoA in matrix.
 Alt: Carnitine acyltransferase I/II and translocase.
Alt: Carnitine acyltransferase I/II and translocase.
Fatty acid oxidation: stages and overview
- Oxidation occurs mainly in the mitochondrial matrix (peroxisomes handle VLCFA > ~22 C).
- Three stages:
- β‑oxidation: successive removal of 2‑carbon units as acetyl‑CoA, producing FADH2 and NADH.
- TCA cycle: acetyl‑CoA oxidation to CO2, producing more NADH/FADH2 and GTP.
- Oxidative phosphorylation: NADH/FADH2 electrons yield ATP.
 Alt: Stage 1 β-oxidation, Stage 2 TCA, Stage 3 electron transport.
Alt: Stage 1 β-oxidation, Stage 2 TCA, Stage 3 electron transport.
The β‑oxidation cycle (4 repeating steps)
- Dehydrogenation by acyl‑CoA dehydrogenase → trans‑Δ2‑enoyl‑CoA; FAD → FADH2.
- Hydration by enoyl‑CoA hydratase → L‑β‑hydroxyacyl‑CoA.
- Dehydrogenation by β‑hydroxyacyl‑CoA dehydrogenase → β‑ketoacyl‑CoA; NAD+ → NADH.
-
Thiolysis by thiolase (acyl‑CoA acetyltransferase) → acetyl‑CoA + acyl‑CoA shortened by 2 C.
-
Overall single-cycle stoichiometry:
\(\(\text{C-acyl-CoA} + \text{FAD} + \text{NAD}^+ + \text{H}_2\text{O} + \text{CoA-SH} \rightarrow \text{C}_{n-2}\text{-acyl-CoA} + \text{FADH}_2 + \text{NADH} + \text{H}^+ + \text{acetyl-CoA}\)\) -
Example: palmitoyl‑CoA (\(C_{16}\)) undergoes 7 cycles → \(8\ \text{acetyl-CoA}\), \(7\ \text{FADH}_2\), \(7\ \text{NADH}\).
- Net ATP yield from complete oxidation of palmitoyl‑CoA ≈ \(108\ \text{ATP}\) (assumes 1.5 ATP/FADH2 and 2.5 ATP/NADH).
 Alt: Pathway from acyl‑CoA to acetyl‑CoA by β-oxidation.
Alt: Pathway from acyl‑CoA to acetyl‑CoA by β-oxidation.
Enzyme complexes and special cases
- Trifunctional protein (TFP): hetero‑octamer handling steps 2–4 for long chains (≥ C12), enabling substrate channeling; defects cause metabolic disease.
 Alt: TFP α and β subunits and activities.
Alt: TFP α and β subunits and activities. - Peroxisomes shorten very‑long chain fatty acids; electrons may reduce O2 (heat, H2O2) rather than feeding respiratory chain.
Energetics (palmitate example)
- Partial summary of ATP yield table (from oxidation of palmitoyl‑CoA to CO2 + H2O):
- From β‑oxidation steps: \(7\ \text{FADH}_2\) and \(7\ \text{NADH}\).
- From oxidation of produced acetyl‑CoA in TCA: additional NADH, FADH2 and GTP per acetyl‑CoA.
- Total ≈ \(108\ \text{ATP}\) per palmitoyl‑CoA (lecture assumption: 1.5 ATP/FADH2; 2.5 ATP/NADH).
 Alt: Table of NADH/FADH2 and ATP yields during palmitoyl-CoA oxidation.
Alt: Table of NADH/FADH2 and ATP yields during palmitoyl-CoA oxidation.
Unsaturated and odd‑chain fatty acids
- Monounsaturated FAs: cis double bonds must be isomerized (Δ3→Δ2) by an isomerase before β‑oxidation proceeds.
- Polyunsaturated FAs: require both an isomerase and a reductase (2,4‑dienoyl‑CoA reductase) for non‑compatible double bonds.
- Odd‑chain FAs: β‑oxidation yields propionyl‑CoA (3 C) instead of acetyl‑CoA; propionyl‑CoA is carboxylated and rearranged to succinyl‑CoA (gluconeogenic).
 Alt: Conversion of propionyl-CoA to succinyl-CoA pathway.
Alt: Conversion of propionyl-CoA to succinyl-CoA pathway.
Role of Coenzyme B12 (cobalamin)
- Methylmalonyl‑CoA mutase (B12‑dependent) converts L‑methylmalonyl‑CoA → succinyl‑CoA during odd‑chain FA catabolism.
- B12 deficiency → impaired propionyl‑CoA metabolism and clinical signs (e.g., pernicious anemia from malabsorption).
Regulation of fatty acid metabolism
- High insulin / high carbohydrate: activate fatty acid synthesis; acetyl‑CoA carboxylase (ACC) is dephosphorylated (active).
- Low glucose / glucagon / epinephrine: phosphorylate ACC (inactive) and activate lipolysis + β‑oxidation.
 Alt: Insulin vs glucagon regulation of ACC and β-oxidation.
Alt: Insulin vs glucagon regulation of ACC and β-oxidation. - Malonyl‑CoA (product of ACC) inhibits carnitine palmitoyltransferase I, preventing simultaneous synthesis and mitochondrial oxidation.
Cellular differences: mitochondria vs peroxisomes/glyoxysomes
- In animals, mitochondrial β‑oxidation funnels electrons via ETF to the respiratory chain → ATP.
- Peroxisomes/glyoxysomes process very‑long/branched chains; in plants, glyoxysomes mobilize seed oils for gluconeogenesis.
 Alt: Mitochondrion vs peroxisome β-oxidation comparison.
Alt: Mitochondrion vs peroxisome β-oxidation comparison.
Ketone bodies: formation and use
- When oxaloacetate is low (e.g., fasting, diabetes), acetyl‑CoA cannot enter TCA and is diverted to ketogenesis in liver mitochondria.
- Ketone bodies: acetoacetate, β‑hydroxybutyrate, and acetone (spontaneous decarboxylation product).
 Alt: Ketone body synthesis pathway (thiolase, HMG‑CoA synthase, HMG‑CoA lyase).
Alt: Ketone body synthesis pathway (thiolase, HMG‑CoA synthase, HMG‑CoA lyase). - Liver makes ketones but lacks β‑ketoacyl‑CoA transferase, so it cannot oxidize them; ketones are exported to other tissues.
- Peripheral tissues (heart, muscle, brain during prolonged fasting) convert ketones back to acetyl‑CoA for TCA oxidation.
Ketones and the brain
- Brain uses glucose normally but adapts to ketone use in prolonged fasting; ketones cross the blood–brain barrier (transporters for monocarboxylates).
- Advantages: ketones supply acetyl‑CoA without requiring FA β‑oxidation in neurons (which consumes lots of O2 and generates ROS).
 Alt: Ketone production by liver and uptake by brain.
Alt: Ketone production by liver and uptake by brain. - Excess ketogenesis can lead to ketoacidosis (dangerous blood pH drop), as seen in uncontrolled diabetes.
Clinical and physiological notes (high‑yield)
- MCAD/LCHAD/TFP deficiencies impair β‑oxidation and present with hypoketotic hypoglycemia and muscle/liver symptoms.
- B12 deficiency impairs odd‑chain FA and certain amino acid catabolism (methylmalonyl‑CoA → succinyl‑CoA).
- Thermogenesis: brown/beige fat and uncoupling in mitochondria increase heat production via FA oxidation.
Quick reference formulas
- Single β‑oxidation cycle (general):
\(\(\text{C-acyl-CoA} + \text{FAD} + \text{NAD}^+ + \text{H}_2\text{O} + \text{CoA-SH} \rightarrow \text{C}_{n-2}\text{-acyl-CoA} + \text{FADH}_2 + \text{NADH} + \text{H}^+ + \text{acetyl-CoA}\)\) - Palmitate (\(C_{16}\)) yields:
\(\(8\ \text{acetyl-CoA},\ 7\ \text{FADH}_2,\ 7\ \text{NADH}\ \Rightarrow\ \approx 108\ \text{ATP (net, lecture values)}\)\)
References: core mechanisms summarized from lecture figures (bile salts, chylomicrons, lipolysis signaling, acyl‑CoA activation, carnitine shuttle, β‑oxidation, propionyl‑CoA pathway, ketogenesis).