Sign up to unlock more features
- Save this deck to your account
- Study flashcards with spaced repetition
- Export to Anki (.apkg) or PDF
- Process documents up to 100 pages
- Images extracted from PDFs and documents
- Better text extraction from your PDFs and documents
- Better flashcards with our more advanced AI model
Flashcards in this deck (38)
-
What is the principle of electrolysis?
Electrolysis is the process of using electrical energy to cause a chemical change, typically to decompose compounds into their elements.
chemistry electrolysis -
In the electrolysis of molten compounds, the compounds are in liquid form, allowing ions to move freely. The positive ions migrate to the cathode and receive electrons, while negative ions move to the anode and give up electrons.
chemistry electrolysis -
During the electrolysis of aqueous sodium chloride, what is produced at the anode?
Chlorine gas (Cl₂) is produced at the anode.
chemistry electrolysis sodium_chloride -
What is produced at the cathode during electrolysis of dilute sulfuric acid?
Hydrogen gas (H₂) is produced at the cathode.
chemistry electrolysis sulfuric_acid -
What is electrolysis?
Electrolysis is the process where a molten ionic compound is broken down by an electric current.
chemistry electrolysis -
Can covalent compounds undergo electrolysis?
No, covalent compounds do not conduct electricity and cannot undergo electrolysis.
chemistry compounds -
Do ionic compounds in the solid state conduct electricity?
No, solid ionic compounds cannot conduct electricity; they lack free ions.
chemistry ionic_compounds -
In solids, ions are fixed in place. When molten or in solution, they are free to move and carry charge.
chemistry ions -
What is an electrode?
An electrode is a rod that conducts electricity into or out of an electrolyte.
chemistry electrolysis -
What is an electrolyte?
An electrolyte is the ionic compound in a molten or dissolved solution that conducts electricity.
chemistry electrolysis -
What is the anode in an electrolysis cell?
The anode is the positive electrode of an electrolysis cell.
chemistry electrolysis -
chemistry ions
-
What is the cathode in an electrolysis cell?
The cathode is the negative electrode of an electrolysis cell.
chemistry electrolysis -
chemistry ions
-
chemistry electrolysis
-
What determines whether a metal or hydrogen is produced at the cathode?
It depends on the reactivity series.
chemistry electrolysis -
chemistry electrolysis
-
chemistry electrolysis
-
chemistry electrolysis
-
What mnemonic helps to remember the electrodes of an electrolytic cell?
The mnemonic is: Positive is Anode, Negative is Cathode (PANIC).
mnemonic chemistry -
chemistry electrolysis
-
chemistry reactivity
-
chemistry electrolysis
-
chemistry electrolysis diagram
-
electrolysis current
-
What carries charge in electrolysis?
Electrons and ions are the charged particles transferring charge.
electrolysis charge -
Where do electrons flow from in electrolysis?
Electrons flow from the negative terminal of the power supply to the cathode.
electrolysis electrons -
What charge does the cathode have?
The cathode gains a negative charge due to the flow of electrons.
electrolysis cathode -
electrolysis electrons
-
At the cathode, cations gain electrons to form atoms. Meanwhile, anions move to the anode and lose electrons.
electrolysis ions -
What happens to electrons after they reach the anode?
Electrons flow back through the external circuit to the positive terminal of the power supply.
electrolysis circuit -
Who are the charge carriers in electrolysis?
- Electrons are the charge carriers in the external circuit.
- Ions are the charge carriers in the electrolyte.
electrolysis carriers -
During electrolysis, electrons flow from the negative terminal to the positive terminal; however, conventional current flows from positive to negative.
electrolysis current -
electrolysis ions
-
electrolysis cathode
-
electrolysis ions
-
electrolysis anode
-
What happens during sodium chloride electrolysis?
Electrons do not flow in the circuit.
Cations move to the cathode and anions move to the anode.
Cations flow freely in the solution.
Only anions gain electrons.
electrolysis sodium_chloride
Electrolysis Principles
- Electrolysis: Process where molten ionic compounds or aqueous solutions are broken down by electric current.
- Conductivity:
- Ionic compounds conduct electricity when molten or in solution, not in solid state.
- Covalent compounds do not conduct electricity.
- Key Terms:
- Electrode: Conducts electricity in/out of electrolyte.
- Electrolyte: Ionic compound in molten/dissolved form that conducts electricity.
- Anode: Positive electrode; attracts anions.
- Cathode: Negative electrode; attracts cations.
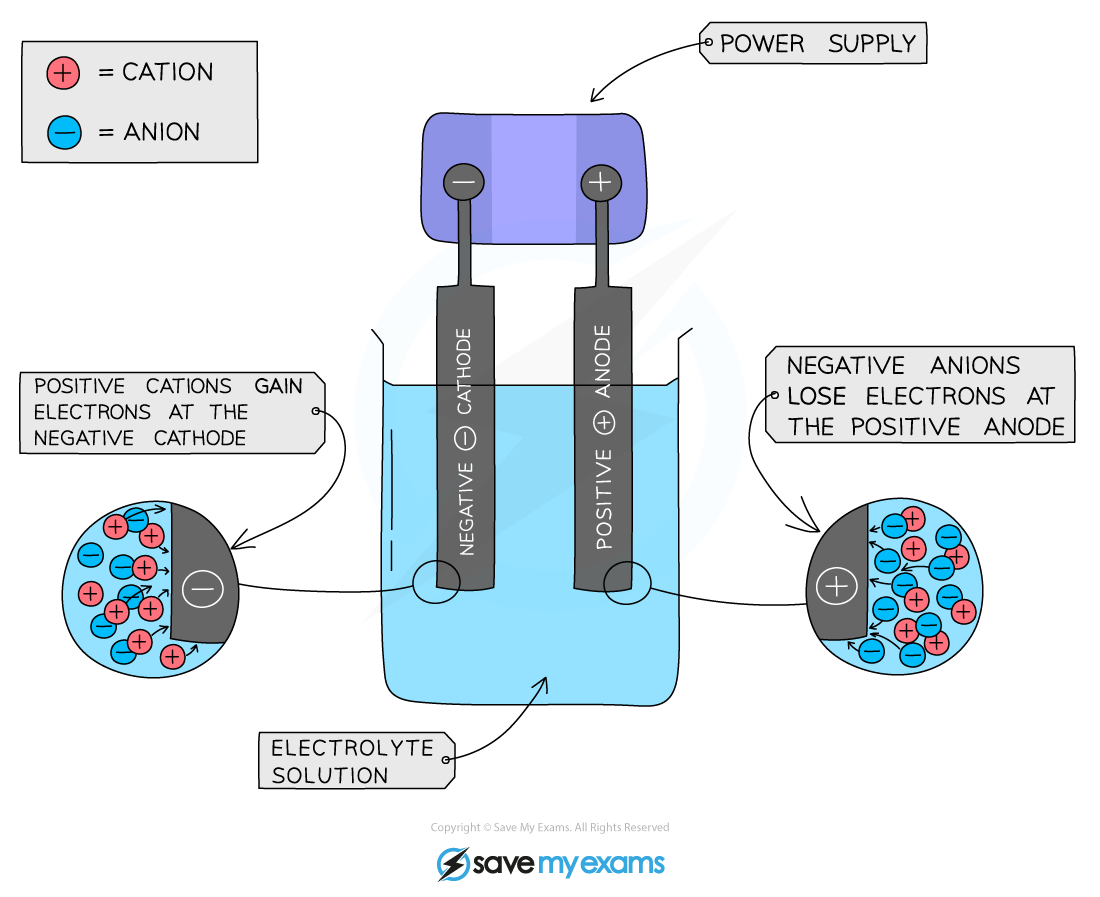
Basic Set-Up of an Electrolytic Cell
- Construction:
- Cations gain electrons at the cathode; anions lose electrons at the anode.
- Metal and Hydrogen Production:
- If metal is less reactive than hydrogen, it is produced at the cathode.
- If more reactive, hydrogen gas is produced instead.
- Non-metals, except hydrogen, are formed at the anode.
- Mnemonic: PANIC (Positive Anode, Negative Is Cathode).
Charge Transfer in Electrolysis
- Movement of Charge:
- Electrons flow from the negative terminal to the cathode, creating a negative charge on the cathode.
- Electrons do not pass through the solution.
- Ionic Migration:
- Cations move to the cathode (reduction: gain of electrons).
- Anions move to the anode (oxidation: loss of electrons).
- Example: Sodium chloride electrolysis illustrates these principles.