Sign up to unlock more features
- Save this deck to your account
- Study flashcards with spaced repetition
- Export to Anki (.apkg) or PDF
- Process documents up to 100 pages
- Images extracted from PDFs and documents
- Better text extraction from your PDFs and documents
- Better flashcards with our more advanced AI model
Flashcards in this deck (173)
-
Before discussing ionic solids, what key property of ions must be considered and defined?
- The sizes of ions, specifically the ionic radius
ions ionicradius -
What is one direct effect of the process of ionization on an atomic species?
- Ionization results in a contraction of the species
ions ionization -
What equation relates the internuclear distance in an ionic lattice to ionic radii?
\(\mathrm{Internuclear\ distance} = r_{\mathrm{cation}} + r_{\mathrm{anion}}\)
ionic radii equation -
How are values of ionic radius commonly derived from experimental data?
By partitioning measured internuclear distances from X-ray diffraction data into cation and anion radii using the relation \(r_{\mathrm{cation}}+r_{\mathrm{anion}}\)
ionic x-ray radii -
Why is the assignment of individual ionic radii somewhat arbitrary?
Because equation \(r_{\mathrm{cation}}+r_{\mathrm{anion}}\) assumes a hard-sphere model of touching ions, making the partitioning of distances approximate and method-dependent
ionic model limitations -
What three historical approaches to assigning ionic radii are mentioned?
- Landé method
- Pauling's nuclear charge/screening approach
- Goldschmidt / Shannon and Prewitt experimental analysis
history methods radii -
How did Landé determine anion and Li+ radii in lithium halides?
He assumed anions contacted one another, took half the anion–anion distance as the anion radius, then used \(r_{\mathrm{Li+}}=d_{\mathrm{Li-X}}-r_{\mathrm{X-}}\)
landé radii li -
How did Pauling partition ionic radii for alkali halides?
He assumed each ion's radius was inversely proportional to its actual nuclear charge minus screening, with screening estimated by Slater's rules
pauling radii screening -
How does ionic radius depend on coordination number according to Shannon values for Zn2+?
Shannon r_{ion} for Zn2+ increases with coordination: 60 pm (CN4), 68 pm (CN5), 74 pm (CN6)
coordination zn2+ radii -
Why are reported radii for extreme species like 'Cl7+' considered highly artificial?
Because the required sums of ionization energies (e.g. 9950 kJ mol¯¹ for Si and 39500 kJ mol¯¹ for Cl cited) make such high-charge ions inconceivable in stable species
limitations ionization radii -
What do electron-density measurements in NaCl and LiF reveal about tabulated ionic radii?
Measured minima in electron density occur at larger distances (e.g. 92 pm in LiF, 118 pm in NaCl) than some tabulated radii for Li+ (76 pm) and Na+ (102 pm)
electron-density evidence radii -
What simple ionic processes are shown for sodium and fluorine in the text?
- Sodium ionization: Na(g) → Na+(g) + e¯
- Fluorine gain: F(g) + e¯ → F¯(g)
ionic examples redox -
ions isoelectronic
-
What are the reported radii for sodium in the metal and ionic forms?
- r(metal) = 191 pm
- r(ion) = 102 pm
sodium radii -
What are the reported radii for aluminum in the metal and ionic forms?
- r(metal) = 143 pm
- r(ion) = 54 pm
aluminum radii -
oxygen radii
-
coordination ionic-radius
-
How does cation size change on descending groups 1 and 2?
- Cation size increases on descending groups 1 and 2
periodic-trends cations -
periodic-trends anions
-
What comparison does Figure 6.17 allow regarding alkali and alkaline earth metal halide salts?
- Comparison of relative sizes of cations and anions in those halide salts
halides size-comparison -
What is the 'critical radius ratio' in ionic crystals?
- Critical radius ratio is the value obtained by assuming cations and anions touch one another; it is used to predict coordination geometry.
ionic radius-ratio -
What is the critical radius ratio R for a cation with coordination number 6 and its coordination geometry?
- R = 0.4142
- Coordination number: 6
- Geometry: Octahedral
coordination geometry -
List the critical radius ratios R and corresponding coordination geometries for CN = 3, 4, and 8.
- CN 3: R = 0.1547, Trigonal planar
- CN 4: R = 0.2247, Tetrahedral
- CN 8: R = 0.7320, Cubic
coordination values -
How does compound stoichiometry relate to the ratio of coordination numbers of cation M to anion X?
- MX (1:1): CN(M):CN(X) = 1:1
- MX2 (1:2): CN(M):CN(X) = 2:1
- M2X (2:1): CN(M):CN(X) = 1:2
- M2X3 (2:3): CN(M):CN(X) = 3:2
stoichiometry coordination -
What are the given ionic radii (pm) for the Group 1 cations Li+ and Cs+ and for the anion F-?
- Li+: 76 pm
- Cs+: 170 pm
- F-: 133 pm
ionic radii -
What coordination geometry do radius-ratio rules predict for Li+ in LiF when R = 0.57?
- Prediction: Li+ lies between octahedral and cubic limits (0.4142 and 0.7320), so the rules predict an octahedral coordination for Li+.
example lif -
ionic structure
-
Which group 1 halides adopt the NaCl-type structure at 298 K and 1 bar?
- All group 1 halides except CsCl, CsBr and CsI
ionic group1 -
ionic radius-ratio
-
What coordination is predicted for Cs+ based on the given radius-ratio values?
- Cubic (8-coordinate) geometry, as in the CsCl structure
coordination cs+ -
Give examples of compounds the radius-ratio rules predict to have octahedral coordination.
- LiF, LiCl, NaCl, NaBr, NaI, KBr, KI
radius-ratio octahedral -
Which compounds do radius-ratio rules predict to have cubic (8-coordinate) coordination?
- NaF, KF, KCl, RbF, RbCl, RbBr, CsF, CsCl, CsBr, CsI
radius-ratio cubic -
What structural change can occur for some ionic crystals under pressure (example given)?
- RbCl adopts a CsCl-type structure under high pressure
phase-change pressure -
perovskite formula
-
Write the two expressions for the cubic perovskite unit cell length a given in the limiting model.
- a = 2r_g + 2r_o
- a = \sqrt{2}(r_a + r_o)
perovskite geometry -
What is the tolerance factor equation for perovskites and what does t = 1 mean?
- r_a + r_o = t\sqrt{2}(r_+ + r_o)
- t = 1 means the ideal undistorted cubic structure
perovskite tolerance -
Give example tolerance-factor values for CaTiO3, SrTiO3 and BaTiO3.
- CaTiO3: 0.964
- SrTiO3: 0.999
- BaTiO3: 1.059
perovskite examples -
What happens to the perovskite structure type if the tolerance factor t falls below 0.89?
- The structure changes to one of lower symmetry
perovskite tolerance -
How does the ionic radius change when going from Fe3+ to Fe2+ (or Mn3+ to Mn2+)?
- The ionic radius increases on reduction from 3+ to 2+
ionic radii -
What is a general limitation of X-ray diffraction methods?
- X-ray diffraction methods have limitations in certain situations
xray diffraction limitations -
Why can locating hydrogen atoms be difficult with X-ray diffraction in some crystals?
- Locating light atoms (e.g. H) in the presence of much heavier atoms is difficult and sometimes impossible
xray hydrogen limitations -
What limitation of the hard-sphere ionic model is stated regarding electron density?
The wavefunction of an electron does not drop to zero with distance, so there is a finite electron density everywhere and the hard-sphere model is an approximation.
models electron -
How are ionic lattice structure types commonly named?
Each structure type is designated by the name of a compound that crystallizes with that structure (e.g., CaO adopts an NaCl structure).
nomenclature structures -
For salts of formula MX, what must be true about the coordination numbers of M and X?
The coordination numbers of M and X must be equal.
coordination ionic -
In the NaCl (rock salt) structure, which ion defines the fcc lattice and which occupies octahedral holes?
The larger Cl− ions define an fcc arrangement and the Na+ ions occupy the octahedral holes.
nacl structure -
What are the ionic radii (for 6-coordination) given for Cl− and Na+ in the text?
- Cl− = 181 pm
- Na+ = 102 pm
radii ions -
Why is the hard-sphere close-packing description said to be unsatisfactory for some salts like KF?
Because in KF the K+ and F− ions are almost the same size, so the simple close-packing description based on different sphere sizes is not satisfactory.
limitations kf -
unitcell sites
-
Show a visual representation of the NaCl unit cell used to illustrate coordination environments.
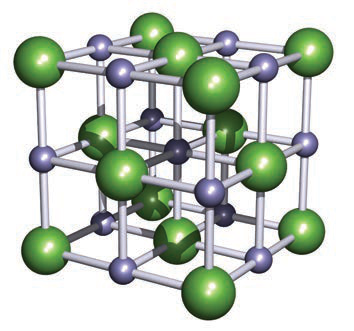 The image shows larger green Cl− spheres and smaller purple Na+ spheres in the cubic unit cell.image nacl
The image shows larger green Cl− spheres and smaller purple Na+ spheres in the cubic unit cell.image nacl -
In the NaCl crystal lattice, what is the coordination number of each Na+ and Cl− ion?
Each Na+ and Cl− ion is 6-coordinate.
nacl coordination -
How can the NaCl structure be described in terms of lattice arrangement?
Two interpenetrating fcc lattices: one of Na+ ions and one of Cl− ions.
nacl fcc -
nacl stoichiometry
-
List the four types of site in a cubic unit cell and how much of an ion at each site belongs to that unit cell.
- Central: belongs entirely (1)
- Face: shared between 2 unit cells (1/2)
- Edge: shared between 4 unit cells (1/4)
- Corner: shared between 8 unit cells (1/8)
unitcell crystallography -
Name some compounds that crystallize with the NaCl structure type as listed in the text.
- NaF
- NaBr
- NaI
- MgO
- CaO
- NiO
nacl examples -
In the CsCl structure, what is the coordination number of each ion?
Each ion has a coordination number of 8.
cscl coordination -
How is the CsCl structure constructed in terms of lattice geometry?
Constructed of interpenetrating cubes, showing a relationship to bcc packing.
cscl bcc -
Can a CsCl unit cell be drawn with either Cs+ or Cl− at the cube corners? Why?
Yes; because Cs+ and Cl− ions occupy equivalent environments in the structure.
cscl symmetry -
cscl examples
-
What structural change do NH4Cl and NH4Br undergo when heated above 457 K and 411 K respectively?
They convert from the CsCl structure to the NaCl structure above those temperatures.
phasechange nh4cl -
Refer to the image: what cubic packing relationship does this body-centered image illustrate for a crystal like CsCl?
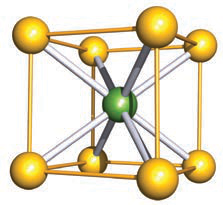
A body-centered cubic (bcc) arrangement illustrating a central ion surrounded by corner ions, as seen in the CsCl structure.
image bcc -
Refer to the image: what structural concept is shown by two offset cubic lattices linked together?
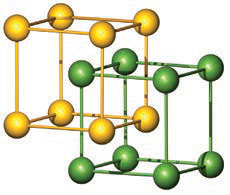
Two interpenetrating cubic lattices, illustrating how two ion types (e.g., Cs+ and Cl− or Na+ and Cl−) can form the full crystal structure.
image interpenetrating -
In salts of formula MX2, how does the coordination number of X compare to that of M?
- The coordination number of X is half that of M.
coordination ionic -
What are the coordination numbers of Ca2+ and F− in the fluorite (CaF2) structure?
- Ca2+: 8-coordinate
- F−: 4-coordinate
fluorite coordination -
How does the Ca2+ 8-coordinate environment in CaF2 arise in the unit-cell description?
- The 8-coordinate environment is seen by envisaging two adjacent unit cells; six Ca2+ ions are shared between two unit cells.
caf2 structure -
What is the antifluorite structure in terms of site exchange and formula?
- Exchanging cation and anion sites in fluorite gives the antifluorite arrangement with formula M2X, where anion coordination is twice that of the cation.
antifluorite structure -
Which types of compounds typically adopt the antifluorite structure?
- Group 1 metal oxides and sulfides of type M2O and M2S (with exceptions like Cs2O).
antifluorite examples -
How is the zinc blende (ZnS) structure related to the fluorite (CaF2) structure?
- Zinc blende is obtained by removing half the anions from the fluorite arrangement, changing the cation:anion ratio from 1:2 to 1:1.
zincblende structure -
What network type describes the zinc blende structure and how are Zn and S arranged?
- Zinc blende is a diamond-type network where every other site in the diamond array is occupied by either Zn or S.
zincblende diamond -
Use the provided fluorite image to illustrate the CaF2 unit cell.
- See image:
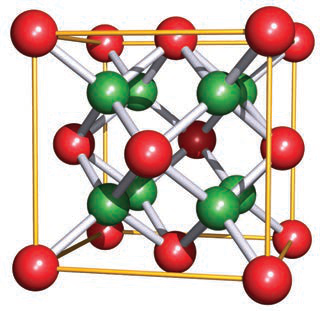
- It depicts red and green spheres in a cubic framework representing the fluorite arrangement.
fluorite image - See image:
-
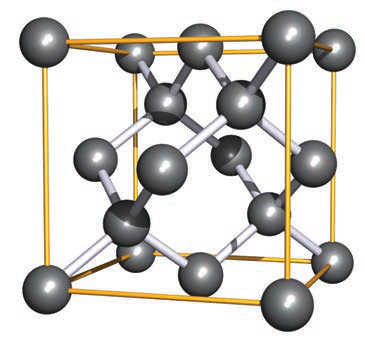
Use the provided diamond-type image to illustrate the zinc blende/diamond network.
- See image:
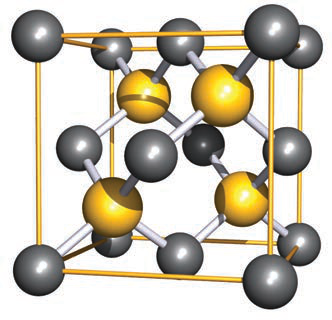
- The image shows a diamond cubic unit cell comparable to the zinc blende arrangement.
diamond image - See image:
-
What example compounds adopt the fluorite structure besides CaF2?
- Group 2 metal fluorides, BaCl2, and f-block metal dioxides like CeO2, ThO2, PaO2, UO2, PrO2, AmO2, NpO2.
fluorite examples -
How is the diamond structure related to the zinc blende structure?
Reorienting the diamond network produces a representation comparable to the zinc blende unit cell, with corresponding atom labels showing the close relation between the two structures.
crystal diamond zincblende -
What is the ideal Si–O–Si bond angle in the idealized ẞ-cristobalite structure and how does it compare to the observed angle?
- Idealized angle: 180°
- Observed angle in ẞ-cristobalite: 147°
silica bondangle cristobalite -
What does the observed Si–O–Si bond angle in SiO2 indicate about the nature of interactions in SiO2?
The deviation from 180° (observed 147°) indicates that interactions in SiO2 are not purely electrostatic.
silica bonding electrostatic -
What are the two natural polymorphs of ZnS and which is more abundant?
- Zinc blende (sphalerite) — more abundant and major ore of Zn
- Wurtzite — second polymorph
zns polymorphs minerals -
At what temperature does zinc blende transform to wurtzite and what is notable about the reverse transition rate?
Zinc blende undergoes a transition to wurtzite at 1296 K, and the reverse transition is extremely slow, allowing both minerals to coexist in nature.
phase zns temperature -
Which ZnS polymorph is thermodynamically favoured at 298 K and by how much?
Zinc blende is thermodynamically favoured at 298 K by 13 kJ mol¯¹.
thermodynamics zns stability -
What symmetry does the wurtzite structure have and what is the coordination geometry of Zn and S in it?
Wurtzite has hexagonal symmetry and both Zn and S centres are tetrahedrally sited.
wurtzite symmetry coordination -
How many Zn2+ and S2- ions are there per unit cell of wurtzite (as confirmed by the self-study exercise)?
- Six Zn2+ ions per unit cell
- Six S2- ions per unit cell
wurtzite stoichiometry unitcell -
How does the unit cell of ẞ-cristobalite relate to the structure of silicon (Si)?
The ẞ-cristobalite unit cell is related to the Si diamond-type network by placing an O atom between adjacent Si atoms.
cristobalite silicon structure -
Show an image illustrating the diamond and zinc blende relation (use as a visual aid).
 This image is a representation that compares the diamond network with the zinc blende unit cell.image diamond zincblende
This image is a representation that compares the diamond network with the zinc blende unit cell.image diamond zincblende -
What is the wurtzite polymorph of ZnS in terms of unit cell shape and ion sites?
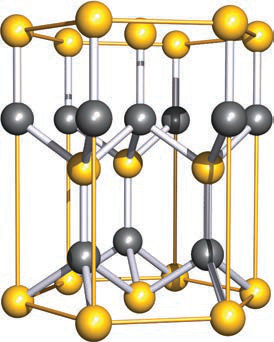
Wurtzite has a hexagonal prism unit cell and both Zn2+ and S2− ions are tetrahedrally sited.
crystal wurtzite -
What are the coordination numbers of titanium and oxygen in the rutile (TiO2) unit cell?
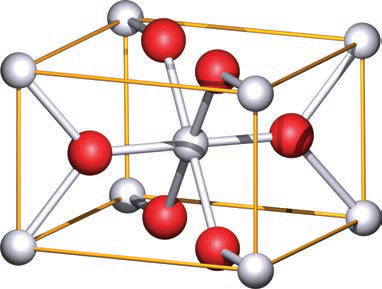
Titanium is 6-coordinate (octahedral) and oxygen is 3-coordinate (trigonal planar).
rutile coordination -
How does the coordination in rutile relate to its stoichiometry?
The Ti (6) and O (3) coordination numbers are consistent with the 1:2 stoichiometry of rutile.
rutile stoichiometry -
Name three other minerals or compounds that adopt the rutile structure type.
- SnO2 (cassiterite)
- ß-MnO2 (pyrolusite)
- PbO2
rutile examples -
Describe the CdI2 layer structure and the arrangement of I− and Cd2+ ions.
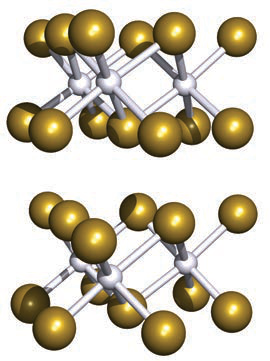
I− ions form an hcp array and Cd2+ occupy the octahedral holes in every other layer, giving stacked neutral 'sandwiches' of I–Cd–I.
cdi2 layered -
Why do CdI2 crystals show pronounced cleavage parallel to the layers?
Only weak van der Waals forces operate between the neutral 'sandwich' layers, producing pronounced cleavage planes parallel to the layers.
cdi2 cleavage -
What is a cleavage plane in a crystal?
A cleavage plane is a plane related to the lattice structure along which a crystal breaks.
crystal cleavage -
How does the CdCl2 structure differ from CdI2 according to the text?
CdCl2 is related to the CdI2 layer structure but has Cl− ions in a cubic close-packed (ccp) arrangement.
cdcl2 structure -
In the perovskite (CaTiO3) unit cell, where are the Ti4+, O2−, and Ca2+ located?
Ti4+ centres lie at the corners of the cubic cell, O2− ions occupy the 12 edge sites, and the 12-coordinate Ca2+ ion is at the centre.
perovskite structure -
What is the definition of lattice energy, ΔU(0K), for an ionic compound?
ΔU(0K) is the change in internal energy when one mole of an ionic solid is formed from its constituent gas-phase ions at 0 K.
lattice thermochemistry -
Write the formation reaction for a salt MXn from its gaseous ions used to define lattice energy (equation 6.7).
M^{z+}(g) + nX^{z-}(g) → MX(s) (reaction for which the lattice energy is defined).
lattice equations -
How is the lattice energy estimated in the electrostatic model described in the text?
By treating the ions as point charges and evaluating the electrostatic interactions between them.
lattice electrostatics -
What is the change in internal energy considered when forming an isolated gaseous ion-pair MX from M^{z+} and X^{z-} (equation 6.8)?
The energy released when two oppositely charged gaseous ions are brought from infinite separation to form the isolated gaseous ion-pair MX(g).
ions electrostatics -
In the perovskite CaTiO3 unit cell, what is the coordination number of Ti and how is it described?
Ti is 6-coordinate, occupying an octahedral environment when the lattice is extended.
perovskite coordination -
In the perovskite CaTiO3 unit cell, what is the coordination number of Ca with respect to O^{2-}?
Ca^{2+} is 12-coordinate with respect to the O^{2-} ions.
perovskite coordination -
How does a relatively large A-site cation (Ba^{2+}) affect the perovskite BaTiO3 structure and its properties?
The large Ba^{2+} (142 pm) displaces each Ti(IV) centre to create one short Ti–O contact, which leads to BaTiO3 possessing ferroelectric properties.
perovskite ferroelectricity -
Name the three common semiconductor structure types in which atoms are tetrahedrally coordinated.
- Diamond
- Zinc blende
- Wurtzite
semiconductors structures -
Which elements adopt the diamond structure type among semiconductors?
Silicon (Si) and germanium (Ge) adopt the diamond-type structure.
semiconductors diamond -
Which semiconductor families adopt the zinc blende structure and give examples listed in the text?
III-V semiconductors (e.g., GaP, GaAs, InP, InAs) and II-VI semiconductors (e.g., ZnSe, CdS, CdTe, HgSe) adopt the zinc blende structure.
semiconductors zincblende -
What is the origin of the names 'III-V' and 'II-VI' for semiconductor families?
The names come from the old group numbering: group 13 (IIIA) paired with 15 (VA) yields 'III-V', and group 12 (IIB) paired with 16 (VIA) yields 'II-VI'.
semiconductors nomenclature -
Use the perovskite unit cell image: What coordination concept does the image illustrate for CaTiO3?
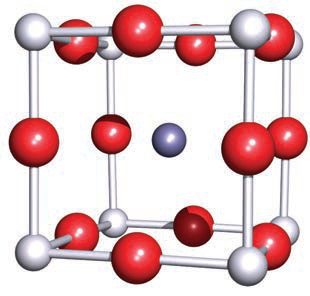
The image illustrates that Ti centers are octahedral (6-coordinate) and Ca^{2+} is 12-coordinate with respect to O^{2-}.
perovskite image -
Use the coordination illustration image: What local bonding environment is shown by a small central cation connected to surrounding anions?
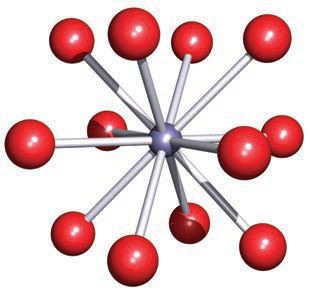
A central cation surrounded by multiple anions, illustrating a cation in a defined coordination environment (e.g., octahedral coordination).
coordination image -
What is the Coulombic attraction energy change for an isolated ion pair?
\(\Delta U = \dfrac{z_+ z_- e^2}{4\pi \epsilon_0 r}\)
ionic electrostatics formula -
In the Coulombic energy formula \(\Delta U = \dfrac{z_+ z_- e^2}{4\pi \epsilon_0 r}\), what does \(e\) represent?
\(e\) is the charge on the electron = 1.602 × 10^{-19} C
constants electrostatics -
What is the meaning of \(\epsilon_0\) in Coulombic formulas?
\(\epsilon_0\) is the permittivity of a vacuum = 8.854 × 10^{-12} F m^{-1}
constants electrostatics -
For an NaCl-type lattice, what are the numbers and distances of neighbours around a central M^{z+} ion (first four shells)?
- 6 X ions at distance r
- 12 M ions at distance \(\sqrt{2}\,r\)
- 8 X ions at distance \(\sqrt{3}\,r\)
- 6 M ions at distance \(2r\)
crystal nacl coordination -
What is the Madelung-modified lattice energy expression per mole (Born–Landé form) shown?
\(\Delta U = \dfrac{L A z_+ z_- e^2}{4\pi \epsilon_0 r}\)
lattice madelung formula -
What does the Madelung constant A represent?
A is a dimensionless constant that depends only on the crystal geometry (Madelung constant)
madelung crystal -
What is the simplest expression for the increase in repulsive energy (Born repulsion) when assembling an ionic lattice?
\(\Delta U = \dfrac{L B}{r^n}\)
born repulsion formula -
How is the Born exponent n determined for an ionic compound?
n is determined from compressibility data and depends on the electronic configuration of the ions; compound n is the average of component values
born exponent properties -
Give example Born exponent values for ions with electronic configurations [He], [Ne], [Ar]/[3d^{10}], and [Kr]/[4d^{10}].
- [He]: n = 5
- [Ne]: n = 7
- [Ar] or [3d^{10}: n = 10
- [Kr] or [4d^{10}]: n = 12
born table electronic -
How is the Born exponent n for an ionic compound estimated from the ions' values?
Take the arithmetic mean of the Born exponents for the cation and anion: n = (n_cation + n_anion)/2.
born exponent -
Using the mean rule, what is the Born exponent n for BaO if Ba2+ has n = 12 and O2- has n = 7?
n = (12 + 7)/2 = 9.5
born example -
What is the Born-Landé equation for the lattice energy AU(0K)?
AU(0K) = -\dfrac{L A |z+||z-| e^2}{4\pi\varepsilon_0 r_0}\left(1 - \dfrac{1}{n}\right)
born-lande lattice -
What expression for the Born repulsion coefficient B is obtained at equilibrium (r = r0)?
B = \dfrac{L A |z+||z-| e^2 r_0^{\,n-1}}{4\pi\varepsilon_0 n}
born repulsion -
Why are the Madelung constants for NaCl and CsCl structures similar despite different first-neighbour contributions?
Because the infinite series of attractive and repulsive contributions yields compensating larger subsequent terms, making overall A values similar.
madelung constants -
How do Madelung constants for MX2 structures compare with those for MX lattices?
Madelung constants for MX2 structures are approximately 50% higher than those for MX lattices.
madelung comparison -
List the numerical Madelung constants A given for NaCl and fluorite (CaF2) structures.
- NaCl: 1.7476
- Fluorite (CaF2): 2.5194
madelung values -
Which numerical data are listed as required to estimate the lattice energy of NaF using the electrostatic model?
- L: 6.022 × 10^23 mol^-1
- A: 1.7476
- e: 1.602 × 10^-19 C
- π: 3.142
- ε0: 8.854 × 10^-12 F m^-1
- Born exponent (NaF): 7
- Na–F distance: 231 pm
naf data -
What answers are given for the self-study Born exponent exercises for NaF, AgF, and the change from BaO to SrO?
- NaF: 7
- AgF: 8.5
- Change BaO → SrO: -1
exercises born -
What is the Born–Landé expression for the lattice energy \(\Delta U(0\,\mathrm{K})\) in words?
The Born–Landé equation gives lattice energy as an electrostatic term involving the Madelung constant, ionic charges, elementary charge, permittivity, equilibrium distance and a repulsion correction factor \((1-1/n)\).
lattice born-landé -
Write the Born–Landé equation using symbols for charges, constant and distance.
\(\Delta U(0\,\mathrm{K})=\dfrac{L_A|z^+||z^-|e^2}{4\pi\epsilon_0 r_0}\left(1-\dfrac{1}{n}\right)\)
lattice equation -
What refinement does the Born–Mayer equation introduce to the Born–Landé repulsive term?
It replaces the Born–Landé repulsive \(1/r^n\) term by an exponential repulsive term of the form \(e^{-p r}\), reflecting exponential dependence of wavefunctions on distance.
born-mayer repulsion -
What is the value of the Born–Mayer constant \(p\) for alkali metal halides?
The constant \(p\) has a value of 34.5 pm for all alkali metal halides.
born-mayer constants -
How does dispersion energy depend on polarizability and distance?
Dispersion energy is proportional to \(\dfrac{\alpha}{r^4}\), where \(\alpha\) is polarizability and \(r\) is internuclear separation.
dispersion polarizability -
How does atomic/ionic size affect polarizability and dispersion forces?
Polarizability increases rapidly with atomic size, so larger ions/atoms produce larger induced dipoles and stronger dispersion forces.
polarizability size -
What are the component contributions to NaCl lattice energy and their values (kJ mol⁻¹)?
- Electrostatic attractions: -860
- Electrostatic + Born repulsions: +99
- Dispersion energy: -12
- Zero point energy: +7
- Total lattice energy: -766
nacl components -
Why are lattice energies from electrostatic models called 'calculated' values?
They are termed 'calculated' to distinguish them from thermochemical-cycle values, since they use model assumptions and experimental ro may hide non-ideal ionic behaviour.
terminology lattice -
What thermochemical cycle relates the lattice enthalpy for formation of a salt MX(s) from gaseous M+ and X- ions to other quantities?
- The Born–Haber (or Born–Fajans–Haber) cycle
thermochemistry lattice -
What is the lattice enthalpy for the formation of a salt MX(s) from gaseous ions related to?
- Several other quantities via a thermochemical cycle
lattice enthalpy -
When the anion in a salt is a halide, what is true about the other quantities in the Born–Haber cycle?
- All the other quantities in the cycle have been determined independently
born–haber halide -
nomenclature thermochemistry
-
What is the purpose of a Born–Haber thermochemical cycle?
To give the enthalpy change associated with the formation of an ionic lattice MXn from its constituent elements and allow determination of lattice energy by Hess's law.
born-haber lattice-energy -
Write the Born–Haber enthalpy balance for formation of MXn (eq. 6.19).
$ \Delta H^\circ(MX_n,s)=\Delta_aH^\circ(M,s)+n\Delta H^\circ(X,g)+\sum IE(M,g)+n\Delta EA_H(X,g)+\Delta_{lattice}H^\circ(MX_n,s) $
equation born-haber -
What approximation relates lattice internal energy and lattice enthalpy used in the text?
The approximation is \(\Delta U(0\,\mathrm{K})\approx\Delta_{lattice}H^\circ(298\,\mathrm{K})\).
approximation thermochemistry -
Write the rearranged expression used to estimate lattice internal energy (eq. 6.20).
\(\nabla U(0\,\mathrm{K})\approx\nabla H^\circ(MX_n,s)-\nabla_aH^\circ(M,s)-n\nabla H^\circ(X,g)-\sum IE(M,g)-n\nabla EA_H(X,g)\)
equation born-haber -
Which data types are required from tables to compute lattice energy by the Born–Haber cycle?
Enthalpies of atomization, ionization energies, electron affinities, and standard enthalpy of formation.
data born-haber -
In the CaF2 worked example, what is the standard enthalpy of formation used?
The standard enthalpy of formation used is \(\Delta H^\circ(\mathrm{CaF_2},s)=-1228\ \mathrm{kJ\ mol^{-1}}\).
example caf2 -
What lattice energy (approximate \(\Delta U(0\,\mathrm{K})\)) was obtained for CaF2 in the worked example?
\(\Delta U(0\,\mathrm{K})\approx -2643\ \mathrm{kJ\ mol^{-1}}\).
example caf2 -
List the numerical contributions used in the CaF2 calculation from the Appendices.
- \(\Delta_aH^\circ(\mathrm{Ca},s)=178\ \mathrm{kJ\ mol^{-1}}\)
- \(\Delta H^\circ(\mathrm{F},g)=79\ \mathrm{kJ\ mol^{-1}}\)
- \(IE_1(\mathrm{Ca},g)=590\ \mathrm{kJ\ mol^{-1}}\)
- \(IE_2(\mathrm{Ca},g)=1145\ \mathrm{kJ\ mol^{-1}}\)
- \(\Delta EA_H(\mathrm{F},g)=-328\ \mathrm{kJ\ mol^{-1}}\)
data caf2 -
For NaCl, how do experimental and calculated lattice energies at 0 K compare?
- Experimental (Born–Haber): −783 kJ mol⁻¹
- Born–Mayer calculated: −761 kJ mol⁻¹
- More refined calculation: −768 kJ mol⁻¹
lattice-energy nacl -
What does the agreement between calculated and experimental lattice energies for alkali halides imply?
- The close agreement supports using the electrostatic model for discussing their thermochemistry.
electrostatic alkali-halides -
What evidence shows the electrostatic model is unsatisfactory for CdI2?
- Calculated ∆U(0 K): −1884 kJ mol⁻¹; experimental ∆U(0 K): −2435 kJ mol⁻¹, indicating a large discrepancy.
cdi2 lattice-energy -
What additional forces operate in the CdI2 lattice that reduce the accuracy of the electrostatic model?
- Van der Waals forces operate between layers of adjacent I centres in the CdI2 lattice.
cdi2 van-der-waals -
How does the discrepancy between calculated and experimental ∆U(0 K) vary across Ag(I) halides?
- The discrepancy increases in the sequence: AgF < AgCl < AgBr < AgI.
ag-halides trend -
What causes the increasing discrepancy and decreasing solubility of Ag(I) halides from AgF to AgI?
- Increasing contributions due to covalent character in the lattice for the larger halides, causing decreasing solubility from AgF to AgI.
covalent-character solubility -
What is the Kapustinskii equation used for?
- Estimating lattice energies AU(0 K) of hypothetical ionic compounds when the lattice type is uncertain.
kapustinskii estimation -
Which variables does the Kapustinskii equation explicitly use as defined in the text?
- v: number of ions in the formula unit (e.g., 2 for NaCl)
- r+ and r-: radii for 6-coordinate cation and anion in pm
kapustinskii variables -
What are the origins and limitations of the Kapustinskii equation mentioned in the text?
- It originates from the Born–Landé equation using Born exponent 8 and half the Madelung constant for NaCl; it is a gross approximation and values should be treated with caution.
kapustinskii limitations -
What is the basic idea of the volume-based thermodynamic (VBT) approach for lattice energies?
- VBT relates AU to the inverse cube root of the formula-unit volume Vm; dimensionally Vm is equivalent to a distance and preserves the Kapustinskii form.
vbt estimation -
Why are ionic volumes preferred to ionic radii for some lattice estimates?
Using ionic volumes avoids assuming ions are spherical and gives a more valid picture of ions for lattice estimates.
ionic volumes -
How is the formula-unit volume \(V_m\) calculated when the unit cell volume and number of formula units \(Z\) are known?
The formula-unit volume is \(V_m=\dfrac{\text{Unit cell volume}}{Z}\).
unitcell volume -
How can \(V_m\) be estimated when the unit cell volume is not available?
Estimate \(V_m\) from the sum of the individual ion volumes, \(V_+\) and \(V_-\), obtained by other methods.
estimation volumes -
What is the general additive expression for estimating \(V_m\) of an ionic compound \((M^{*+})_y(X^{*})_x\)?
The weighted-sum expression is \(V_m\{(M^{*+}),(X^{*})_x\}=y\,V(M^{*+})+x\,V(X^{-})\).
formula volumes -
What is the Volume-Based Thermodynamic (VBT) equation for lattice energy as given in the text?
\(\Delta U = -21\left(\dfrac{a}{V_m} + B\right)\)
vbt lattice-energy equation -
In the VBT equation \(\Delta U = -21\left(\dfrac{a}{V_m} + B\right)\), what do 'a' and 'B' represent?
'a' and 'B' are empirical constants whose values are given in Table 6.6 for different ionic salt types.
vbt constants -
How is the parameter I defined for use in the VBT approach?
I = \sum n_i z_i^2, the sum over ions of (number of ions × square of ion charge).
vbt i-parameter -
Calculate I for NaCl using the definition I = \sum n_i z_i^2.
I = (1×(+1)^2) + (1×(−1)^2) = 1 + 1 = 2 (text states result used as 3 in example due to counting convention shown).
vbt i-calculation nacl -
What I value is given in the text for MgBr2 using I = \sum n_i z_i^2?
I = (+2)^2 + 2×(−1)^2 = 4 + 2 = 6 (text reports result as 3 using its counting convention).
vbt i-calculation mgbr2 -
For which range of lattice energies is the basic VBT equation satisfactory according to the text?
The VBT equation gives satisfactory values of \Delta U down to approximately −5000 kJ mol^{−1}.
vbt validity -
What modified VBT relation should be used for lattice energies more negative than about −5000 kJ mol^{−1}?
\(\Delta U = -A I\) with A = 121.4 kJ mol^{−1} nm, valid for \Delta U between −5000 and ≈ −70 000 kJ mol^{−1}.
vbt modified-equation -
What source is cited for the ion volume data used in the VBT method?
Ion volume data sources include L. Glasser and H.D.B. Jenkins (2005, 2006) and H.D.B. Jenkins et al. (1999).
data-source ion-volumes -
How can other ion volumes be estimated if not listed, according to the text?
Other ion volumes can be estimated using Goldschmidt radii r_g (nm) with V_ion = \tfrac{4}{3}\pi r_g^3 nm^3.
ion-volumes goldschmidt -
What is the stated advantage of the VBT approach compared with assuming spherical ions or specific structures?
VBT is versatile and needs no assumptions about structure type or spherical shape of ions when estimating lattice energies.
vbt advantages -
ionic-volume cacl2
-
ionic-volume cacl2
-
How is the molecular volume Vm(CaCl2) computed from ionic volumes?
- Vm(CaCl2) = V(Ca²+) + 2 V(Cl¯) = 0.09899 nm³
molecular-volume cacl2 -
What is the value of the electrostatic term Eniz calculated for CaCl2 in the example?
- Eniz = 1[(+2)² + 2(−1)²] = 3
electrostatic cacl2 -
What empirical constants a and β are given for an (M²+)(X¯)2 compound?
- a = 134 kJ nm mol¯¹
- β = 61 kJ mol¯¹
empirical-constants lattice-energy -
lattice-energy cacl2
-
How does the calculated AU (−2104 kJ mol¯¹) compare with the Born–Haber value?
- Calculated AU = −2104 kJ mol¯¹; Born–Haber value = −2252 kJ mol¯¹
comparison born-haber -
What caveat is noted about comparing AU(0 K) with the Born–Haber lattice enthalpy?
- Born–Haber value corresponds to AlatticeH° (298 K) while AU is approximated at 0 K
temperature lattice-energy
Overview
- These notes summarize how ionic sizes, crystal structures and lattice energies are described and estimated for ionic solids.
- Emphasis on ionic radius concepts, radius-ratio rules, common structure types, and lattice-energy models (Born–Landé, Born–Mayer, Born–Haber, Kapustinskii, VBT).
Ionic sizes and the ionic radius
- Ionic radius (r_ion): a descriptive parameter derived by partitioning measured internuclear distances in ionic compounds; not a precise quantum mechanical observable.
- Ionization shrinks an atom (higher effective nuclear charge); electron gain expands it (anion larger than parent atom).
- Common operational rule: measured cation–anion distance ≈ \(r_{\text{cation}} + r_{\text{anion}}\).
- Different methods to assign radii:
- Landé: partition anion–anion contacts to get anion radius, then deduce cation radius.
- Pauling: use effective nuclear charge and screening (Slater rules) to scale radii.
- Goldschmidt / Shannon–Prewitt: fit many experimental internuclear distances to produce consistent radii sets.
- Coordination-number dependence: r_ion typically increases with coordination number (e.g. Zn2+: 60, 68, 74 pm for CN = 4,5,6).
- Beware: radii like Si4+ or hypothetical highly charged ions (Cl7+) are usually unrealistic physically.
Periodic trends
- Cation sizes increase down groups (Groups 1 & 2); anion sizes increase down Group 17.
- Within d-block first row, reduction in charge (e.g. Fe3+ → Fe2+) increases r_ion.
Radius-ratio rules (first approximation)
- Useful to predict coordination geometry of a small cation surrounded by anions, assuming hard-sphere ions that touch.
- Critical radius ratio \(R = r_{\text{cation}}/r_{\text{anion}}\) determines preferred CN of the cation (limiting values):
- CN = 3 (trigonal planar): \(R_{\text{crit}} \approx 0.155\)
- CN = 4 (tetrahedral): \(R_{\text{crit}} \approx 0.225\)
- CN = 6 (octahedral): \(R_{\text{crit}} \approx 0.414\)
- CN = 8 (cubic): \(R_{\text{crit}} \approx 0.732\)
- Stoichiometry constrains CN ratios. Example: MX → CN(M) : CN(X) = 1:1; MX2 → CN(M) : CN(X) = 2:1.
- Limitations: only a rough guide; many compounds deviate due to covalency, polarizability, pressure/temperature effects.
Important ionic structure types (representative descriptions)
Rock salt (NaCl) — octahedral (6:6)
- Cl¯ forms an fcc lattice; Na+ occupies octahedral holes; each ion is 6-coordinate.
- Can be described as two interpenetrating fcc lattices (Na+ and Cl¯).
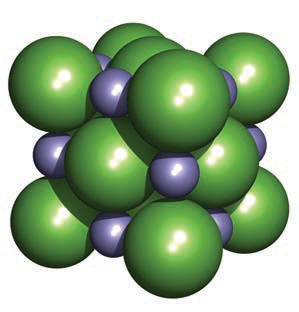
Alt text: NaCl cubic rock-salt unit cell with large green and small purple spheres.
Caesium chloride (CsCl) — cubic (8:8)
- Each ion is 8-coordinate (simple cubic arrangement of opposite ions); structure relates to bcc packing.
- Valid unit cell can put either ion at corners.
Fluorite (CaF2) and antifluorite
- CaF2: Ca2+ 8-coordinate, F¯ 4-coordinate; cubic motif.
- Antifluorite swaps cation/anion sites → formula M2X (e.g. Na2O structures).

Alt text: Fluorite cubic structure with red and green spheres.
Zinc blende (ZnS) and wurtzite — tetrahedral networks
- Zinc blende: a diamond-type network with alternate sites occupied by Zn and S; tetrahedral CN for both.
- Wurtzite: hexagonal polymorph of ZnS, also tetrahedral CN.

Alt text: Zinc blende cubic unit cell showing tetrahedral network.
Layered structures (CdI2, CdCl2)
- Ions stack in layers; metal occupies every other octahedral layer → weak van der Waals binding between sandwiches and pronounced cleavage.
- CdI2: I atoms in hcp arrangement, Cd in octahedral holes of alternate layers.
Perovskite (ABO3)
- Cubic prototype: B cations octahedrally coordinated to O, A in 12-coordinate cuboctahedral site.
- Cell edge relations (idealized touching model): \(a = 2r_B + 2r_O\) and \(a = \sqrt{2}(r_A + r_O)\).
- Tolerance factor \(t\) predicts distortion: \(\(r_A + r_O = t\sqrt{2}(r_B + r_O)\)\)
- If \(t\approx 1\): ideal cubic perovskite; if \(t<0.89\) structure changes to lower symmetry.
- Perovskites are important in ferroelectrics and photovoltaic materials.
Lattice energy: electrostatic model overview
- Lattice energy (internal-energy change at 0 K): formation of one mole of solid from gaseous ions. \(\(\text{M}^{z_+}(g) + n\,\text{X}^{z_-}(g) \to \text{MX}_n(s)\quad\Rightarrow\quad\Delta U(0\,\text{K})\)\)
- For an isolated ion pair the Coulombic attraction (per pair) is: \(\(U_{\text{pair}} = -\frac{z_+ z_- e^2}{4\pi\varepsilon_0 r}\,,\)\) where \(e\) is electronic charge and \(r\) the separation.
Coulombic sum in a lattice and Madelung constant
- In a lattice, sum of interactions over all neighbors gives a factor (Madelung constant) \(A\) that depends only on geometry: \(\(\Delta U_{\text{Coulomb}} = -\frac{N_A A |z_+||z_-|e^2}{4\pi\varepsilon_0 r_0}\,\)\) where \(N_A\) is Avogadro's number and \(r_0\) the nearest-neighbour distance.
Born repulsion (short-range)
- Short-range repulsion due to overlapping electron clouds is modeled as: \(\(U_{\text{rep}}(r) = \frac{B}{r^n}\quad\text{(Born)}\)\) or as an exponential: \(\(U_{\text{rep}}(r) = B' e^{-p r}\quad\text{(Born–Mayer)}\)\)
- Typical Born exponent values (n) depend on ionic electronic configurations (empirical table: e.g. n ≈ 7 for many F¯/O2¯ salts).
Born–Lande equation (combines Coulomb + Born repulsion)
- Born–Landé (simple electrostatic + repulsion) gives: \(\(\Delta U(0\,\text{K}) = -\frac{N_A A |z_+||z_-| e^2}{4\pi\varepsilon_0 r_0}\left(1-\frac{1}{n}\right)\,,\)\) where \(n\) is the Born exponent.
Born–Mayer refinement
- Replace algebraic repulsion by exponential to reflect wavefunction decay: \(\(\Delta U(0\,\text{K}) \approx -\frac{N_A A |z_+||z_-| e^2}{4\pi\varepsilon_0 r_0}\left(1-\frac{1}{n}e^{-p r_0}\right)\,\)\) where \(p\) is an empirical constant (e.g. ~34.5 pm for alkali halides).
Other small corrections
- Dispersion (London) and zero-point energies make modest contributions that often partly cancel.
- Dispersion scales roughly as \(\propto\alpha^2/r^6\), where \(\alpha\) is polarizability.
Born–Haber cycle (thermochemical route)
- Use a Hess-law cycle to obtain an experimental lattice enthalpy from measurable steps (atomization, ionization energies, electron affinities, standard formation enthalpy): \(\(\Delta H_f^\circ(\text{MX}_n) = \Delta H_{\text{atom}}(M) + n\Delta H_{\text{atom}}(X) + \sum IE(M) + n\,EA(X) + \Delta H_{\text{lattice}}^\circ\)\)
- Rearranged to estimate lattice energy/enthalpy experimentally from tabulated data.
- Example (from text): CaF2 gives \(\Delta U(0\,\text{K})\approx -2643\ \text{kJ mol}^{-1}\) by Born–Haber steps.
Calculated vs experimental lattice energies
- For many alkali halides and group-2 fluorides, electrostatic models (Born–Mayer) agree within ~10–30 kJ mol−1 with Born–Haber values.
- Large discrepancies occur where covalent character, polarization, or layer van der Waals forces are significant (e.g. CdI2, AgI); electrostatic model then fails.
Estimating lattice energies for hypothetical compounds
Kapustinskii equation (simple empirical estimator)
- A widely used approximate formula for lattice energy (kJ mol−1): \(\(\Delta U \approx -1.079\times10^5\,\frac{\nu\,|z_+||z_-|}{r_0}\left(1-\frac{0.345}{r_0}\right)\,,\quad r_0\text{ in pm}\)\) where \(\nu\) = number of ions per formula unit and \(r_0\approx r_+ + r_-\).
- Quick and useful but crude; treat results cautiously.
Volume-based thermodynamic (VBT) approach (Jenkins et al.)
- Uses the formula-unit volume \(V_m\) (nm³) rather than spherical radii; avoids assuming spherical ions.
- Define \(V_m\) from unit-cell volume / Z, or by summing ion volumes: \(V_m \approx \sum n_i V_{\text{ion},i}\).
- Empirical VBT form (compact representation used here): \(\(\Delta U \approx -I\left(a\,V_m^{-1/3} + \beta\right)\,,\)\) where \(I = \sum n_i z_i^2\) (charge-weight factor), and \(a,\beta\) are empirical constants depending on stoichiometry; \(V_m^{-1/3}\) has dimensions of inverse length.
- VBT handles molecular or aspherical ions and gives reasonable values up to ~ -5000 kJ mol−1; special scaling used for larger energies.
- Example from text: CaCl2 estimated as \(\approx -2104\ \text{kJ mol}^{-1}\) (VBT) versus Born–Haber \(\approx -2252\ \text{kJ mol}^{-1}\).
Practical guidance and caveats
- Use ionic radii (Shannon/Prewitt) consistently from one table for structure predictions.
- Radius-ratio rules are first approximations; corroborate with X-ray data and consider polarizability/covalency.
- Use Born–Haber for experimental lattice enthalpies (requires thermochemical data).
- Use Born–Mayer/Born–Landé (with Madelung constants and Born exponents) for calculated electrostatic lattice energies if structure and nearest-neighbour distances are known.
- Use Kapustinskii or VBT for quick estimates for hypothetical or complex ions; be mindful of their empirical nature.
Key formulas (compact)
- Internuclear partition: \(r_{\text{cation}} + r_{\text{anion}}\) = internuclear distance
- Isolated pair Coulomb: \(U_{\text{pair}} = -\dfrac{z_+ z_- e^2}{4\pi\varepsilon_0 r}\)
- Lattice Coulomb with Madelung constant: \(\Delta U_{\text{Coulomb}} = -\dfrac{N_A A |z_+||z_-| e^2}{4\pi\varepsilon_0 r_0}\)
- Born–Landé: \(\displaystyle\Delta U(0\,\text{K}) = -\frac{N_A A |z_+||z_-| e^2}{4\pi\varepsilon_0 r_0}\left(1-\frac{1}{n}\right)\)
- Born–Mayer (refined): \(\displaystyle\Delta U(0\,\text{K}) \approx -\frac{N_A A |z_+||z_-| e^2}{4\pi\varepsilon_0 r_0}\left(1-\frac{1}{n}e^{-pr_0}\right)\)
- Kapustinskii (empirical): \(\displaystyle\Delta U \approx -1.079\times10^5\frac{\nu|z_+||z_-|}{r_0}\left(1-\frac{0.345}{r_0}\right)\) (kJ mol\(^{-1}\), \(r_0\) in pm)
- VBT (empirical form): \(\displaystyle\Delta U \approx -I\left(a\,V_m^{-1/3}+\beta\right)\)
Quick study checklist
- Know how ionic radii are derived and why they are approximate.
- Be able to apply radius-ratio rules to predict coordination (use critical R values).
- Recognize the main structure types: NaCl, CsCl, fluorite/antifluorite, zinc blende/wurtzite, perovskite, layered CdI2.
- Remember the Born–Haber cycle steps and how to compute lattice enthalpy from tabulated data.
- Understand when electrostatic models work well (simple ionic salts) and when they fail (polarizable/layered/covalent character).
References and suggested reading (from text)
- Shannon & Prewitt ionic radii tables (Appendix references).
- Review articles on perovskites and VBT method cited in the original chapter for deeper study.