Sign up to unlock more features
- Save this deck to your account
- Study flashcards with spaced repetition
- Export to Anki (.apkg) or PDF
- Process documents up to 100 pages
- Images extracted from PDFs and documents
- Better text extraction from your PDFs and documents
- Better flashcards with our more advanced AI model
Flashcards in this deck (386)
-
What is the basic structure of proteins?
Proteins are built from amino acids and formed through peptide bonds.
biology proteins -
How are proteins digested and absorbed in the body?
Proteins are broken down into amino acids by enzymes in the stomach and small intestine, then absorbed into the bloodstream.
biology digestion proteins -
biology proteins functions
-
What are the health impacts of too much protein?
Excess protein can lead to kidney damage, dehydration, and increased heart disease risk.
health nutrition proteins -
What are the health impacts of too little protein?
Insufficient protein can cause muscle wasting, weakened immunity, and protein deficiencies.
health nutrition proteins -
What is a protein deficiency?
A protein deficiency occurs when the body does not get enough protein to meet its needs, leading to health issues.
health nutrition proteins -
What are protein allergies?
Protein allergies occur when the immune system mistakenly identifies a protein as harmful, leading to allergic reactions.
health nutrition allergies -
biology proteins
-
biology proteins
-
biology proteins
-
biology amino_acids
-
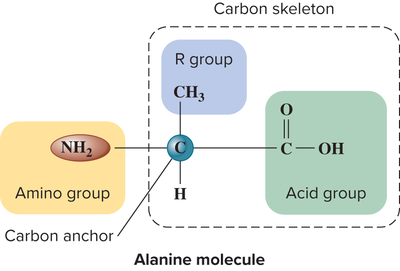
What is the structure of an amino acid?
An amino acid contains a central carbon atom bound to a hydrogen atom, an amino group, a carboxylic acid group, and an R group (side chain).
biology amino_acids -
How many different amino acids are found in the human body?
There are 20 different amino acids found in the proteins of the human body.
biology amino_acids -
biology amino_acids
-
The structure of an alanine molecule includes a central carbon atom bound to: - hydrogen atom - amino group (NH2) - acid group (COOH) - R group (CH3).
biology alanine -
What is represented as R group in amino acids?
The R group in amino acids is the side chain that varies among different amino acids.
biology amino_acids -
What does the carbon skeleton refer to in amino acids?
The carbon skeleton refers to the central carbon atom and its bonds in an amino acid structure.
biology amino_acids -
biology alanine
-
biology amino_acids
-
What are conditionally essential amino acids?
Amino acids that are normally nonessential but become essential under certain conditions.
biology amino_acids -
Name one essential amino acid.
- Histidine
- Isoleucine
- Leucine
- Lysine
- Methionine
- Phenylalanine
- Threonine
- Tryptophan
- Valine
biology amino_acids -
Name one nonessential amino acid.
- Alanine
- Aspartic acid
- Asparagine
- Glutamic acid
- Serine
- Arginine*
- Cysteine*
- Glutamine*
- Glycine*
- Proline*
- Tyrosine*
biology amino_acids -
Which amino acids can become essential under certain conditions?
- Arginine
- Cysteine
- Glutamine
- Glycine
- Proline
- Tyrosine
biology amino_acids -
What process allows the body to synthesize nonessential amino acids when they are not available from the diet?
Transamination
biochemistry amino_acids -
biochemistry amino_acids
-
biochemistry amino_acids
-
What are the products of the transamination reaction between alanine and alpha-ketoglutarate?
Pyruvate and glutamate
biochemistry amino_acids -
The reaction of transamination can be summarized as: Alanine + α-Ketoglutarate <=> Pyruvate + Glutamate.
biochemistry amino_acids -
biochemistry structures
-
biochemistry structures
-
biochemistry structures
-
biochemistry structures
-
nutrition amino_acids
-
nutrition amino_acids
-
What are some conditions that can make amino acids conditionally essential?
- Premature infancy
- Illness or stress
- Periods of rapid growth
- Aging
nutrition amino_acids -
What are amino acid derivatives?
Chemically modified versions of amino acids that contain nitrogen and have important physiological roles.
biochemistry aminoacids -
neurotransmitters serotonin
-
neurotransmitters serotonin
-
neurotransmitters dopamine
-
neurotransmitters dopamine
-
hormones tyrosine
-
What is the function of thyroxine (T4) and triiodothyronine (T3)?
Involved in regulating metabolism.
hormones metabolism -
hormones melatonin
-
hormones sleep
-
energy creatine
-
What is the function of creatine?
Forms phosphocreatine storage in muscle cells for quick energy during short bursts of activity.
energy muscle -
energy carnitine
-
energy fattyacids
-
immune histamine
-
What functions does histamine serve?
Mediator of allergic reactions, involved in immune responses and gastric acid secretion.
immune allergy -
pigment melanin
-
pigment color
-
signaling nitricoxide
-
What role does nitric oxide play?
Helps regulate blood vessel dilation, neurotransmission, and immune responses.
signaling bloodvessels -
nutrition protein
-
nutrition protein
-
When do protein needs increase?
During pregnancy, breastfeeding, rapid growth, recovery from illness, blood losses, and burns.
nutrition protein -
What are complete proteins?
Food sources that contain adequate proportions of all essential amino acids.
nutrition protein -
What is the primary source of protein for most of the world's population?
Most of the world gets protein from plants.
nutrition protein -
What do animal products provide that plant sources typically do not?
Animal products provide B vitamins and minerals but are low in fiber.
nutrition protein -
nutrition protein
-
What is a method to achieve complete protein intake from plants?
Complementary combinations of certain plant foods.
nutrition protein -
Rice provides methionine (Met) and cysteine (Cys), while beans are high in lysine (Lys). Combined, they provide all essential amino acids as in Rice and beans.
nutrition protein -
What are the potential downsides of animal protein sources?
They can be high in saturated fat and cholesterol.
nutrition protein -
What is a peptide bond?
A peptide bond is a chemical bond that links amino acids together via the acid group of one amino acid and the amino group of the next one.
biochemistry proteins -
biochemistry proteins
-
biochemistry proteins
-
What is the primary structure of a protein?
The primary structure is a linear chain of amino acids linked by peptide bonds.
biochemistry protein_structure -
What is the secondary structure of a protein?
The secondary structure involves the coiling of a polypeptide chain.
biochemistry protein_structure -
What is the tertiary structure of a protein?
The tertiary structure is the three-dimensional structure, including interactions between various amino acid groups on the chain.
biochemistry protein_structure -
What is the quaternary structure of a protein?
The quaternary structure is made up of two or more polypeptide chains arranged in a specific way.
biochemistry protein_structure -
What is an example of a protein with quaternary structure?
Hemoglobin, a protein in red blood cells made up of four polypeptide chains.
biochemistry hemoglobin -
The four levels of protein structure are: (a) Primary structure (b) Secondary structure (c) Tertiary structure (d) Quaternary structure.
biochemistry protein_structure -
biochemistry proteins
-
biochemistry proteins
-
The process of protein synthesis involves bonding individual amino acids to form a polypeptide chain which then folds into a 3D shape.
biochemistry protein_synthesis -
What is the mutation that occurs at position 6 of the beta-globin protein?
Glutamic acid (Glu) is replaced by Valine (Val), known as Glu6Val or E6V.
biology genetics -
How does the shape of normal red blood cells differ from sickle-shaped cells?
Normal red blood cells are disc-shaped, while sickle-shaped cells have long chains of molecules.
biology cell_biology -
The mutation Glu6Val results in the alteration of the hemoglobin shape and can cause sickle-shaped red blood cells, leading to changes in function. The normal shape is disc-shaped, while the altered shape is sickle-shaped.
biology genetics -
What is the effect of altered protein shape on function?
Altered protein shape may lead to altered function.
biology proteins -
What are the two shapes compared in the study of hemoglobin?
Normal hemoglobin and sickle-shaped hemoglobin.
biology hemoglobin -
What visual representation is used to compare normal and sickle-shaped red blood cells?
A comparison image showing the progression from normal to sickle-shaped cells.
biology cell_biology -
biology gene_expression
-
biology cell_biology
-
biology gene_expression
-
biology translation
-
biology translation
-
biology translation
-
biology proteins
-
biology gene_expression
-
biology cell_biology
-
biology gene_expression
-
biology translation
-
biology translation
-
biology translation
-
biology illustration
-
What are the stages where gene expression for proteins can be controlled?
- Transcription
- Translation
- Post-translational modification
biology gene_expression -
What does transcriptional regulation involve?
Regulating initiation and progression of mRNA synthesis, e.g. epigenetic modifications, transcription factors.
biology transcription -
What is post-transcriptional regulation?
Regulating mRNA after it has been produced, e.g. microRNAs.
biology post-transcriptional -
biology translation
-
biology post-translational
-
nutrition gene_expression epigenetics
-
Which transcription factor is regulated by glucose?
Carbohydrate Response Element Binding Protein (ChREBP)
nutrition gene_expression transcription_factors -
nutrition gene_expression transcription_factors
-
nutrition gene_expression transcription_factors
-
nutrition gene_expression translation
-
nutrition gene_expression post-translational_modifications
-
What regulates insulin gene transcription?
A promoter region that serves as a binding site for transcription factors.
biology insulin transcription -
Which transcription factor is associated with low glucose conditions?
Hdac interacts with Pdx-1, inhibiting insulin transcription.
biology insulin glucose -
What happens to Pdx-1 in high glucose conditions?
Set7/9 and p300 co-activate Pdx-1, promoting insulin transcription.
biology insulin glucose -
biology insulin pdx1
-
biology insulin glucose
-
biology insulin glucose
-
What is the source of the information regarding insulin gene transcription?
Biochemical Journal (2008) 415, 1-10 - Sreenath S. Andrali, Megan L. Sampley and others.
biology insulin source -
What is illustrated in the diagram regarding insulin gene expression?
Regulation by Pdx-1 in response to glucose levels.
biology insulin diagram -
biochemistry insulin
-
biochemistry insulin
-
biochemistry insulin
-
biochemistry insulin
-
biochemistry insulin
-
biochemistry insulin
-
What does the diagram illustrate about insulin processing?
The conversion of preproinsulin to active insulin
biochemistry insulin -
biology proteins
-
What is the function of enzymes?
They speed up metabolic reactions. Examples: amylase, pepsin, PFK1.
biology enzymes -
biology transport
-
biology immunity
-
biology muscle
-
biology hormones
-
biology regulation
-
biology proteins
-
biology enzymes
-
biology transport
-
biology hormones
-
biology muscle
-
biology immunity
-
biology regulation
-
biology proteins
-
biology proteins
-
What are common food preparation methods that cause protein denaturation?
- Cooking
- Whipping
- Addition of alcohol or acids
cooking food_science -
How does hydrochloric acid affect food proteins in the stomach?
It denatures them, making them easier to digest.
biology digestion -
What occurs in the stomach during protein digestion?
Proteins undergo denaturation by stomach acid and partial digestion by pepsin.
biology digestion -
What enzymes does the pancreas secrete in the small intestine for protein digestion?
The pancreas secretes trypsin and chymotrypsin.
biology digestion -
Where does final digestion of proteins occur?
Final digestion occurs within absorptive cells in the small intestine.
biology digestion -
What is the role of hydrochloric acid (HCl) in protein digestion?
HCl denatures food proteins and converts inactive pepsinogen to active pepsin.
biology digestion -
What do trypsin and chymotrypsin break down polypeptides into?
They break down polypeptides into shorter peptides and amino acids.
biology digestion -
nutrition calories
-
What is highlighted in the illustration of the human digestive system?
The illustration shows the role of protein digestion in the stomach, small intestine, and absorptive cells.
biology digestion -
biology proteins digestion
-
biology proteins absorption
-
biology proteins absorption
-
Where do amino acids go after entering the capillary of the villus?
They travel to the liver via the hepatic portal vein.
biology proteins circulation -
biology proteins liver
-
biology proteins digestion
-
biology proteins absorption
-
biology proteins transport
-
What is the role of pancreatic enzymes in protein digestion?
They break down proteins into short peptides.
biology proteins digestion -
What is depicted in the diagram related to protein absorption?
Protein digestion and absorption in the gut.
biology proteins visual -
What is the source of the diagram illustrating protein digestion?
<a href="https://hel1.your-objectstorage.com/anki-decks/flashcard_media/anki_decks/anki_decks/09-10_Proteins_F_m3BHq4F.pdf_13.png">Diagram</a>
biology proteins visual -
nutrition amino_acids
-
nutrition amino_acids
-
nutrition amino_acids
-
What is the amino acid pool?
A reservoir of amino acids available for protein synthesis and other processes
nutrition amino_acids -
nutrition amino_acids
-
nutrition amino_acids
-
What is protein turnover?
A cellular process of breaking down proteins and recycling their amino acids
nutrition amino_acids -
The total daily protein turnover is large, with 2/3 of the amino acids in the amino acid pool coming from recycled proteins and 1/3 from dietary proteins.
nutrition amino_acids -
What can the amino acid pool be used for?
- Energy
- Synthesis of glucose or fatty acids
- Synthesis of nonprotein molecules containing nitrogen
nutrition amino_acids -
What is depicted in the flowchart about dietary proteins?
The fate of dietary proteins, showing digestion into an amino acid pool and its uses
nutrition amino_acids -
What happens to excess amino acids in the body?
They undergo deamination, where the amino group is removed, forming ammonia (NH3).
biology amino_acids -
What is formed from ammonia (NH3) in the liver?
Urea, a waste product of amino acid metabolism, is formed from ammonia in the liver.
biology metabolism -
biology excretion
-
biology amino_acids
-
biology metabolism
-
biology excretion
-
What is the potential waste product of amino acid metabolism?
Ammonia (NH3) is a potentially toxic waste product formed during deamination.
biology toxicology -
What is the role of ATP in urea synthesis?
ATP is used in the liver to convert ammonia (NH3) and CO2 into urea.
biology metabolism -
What is transported to the liver as glutamate during deamination?
The amino group (NH2) is transported to the liver as glutamate.
biology amino_acids -
What toxic waste product is formed during the deamination of amino acids?
Ammonia (NH3) is formed during the deamination of amino acids.
biology toxicology -
biology diagrams
-
health biochemistry
-
health biochemistry
-
health biochemistry
-
health biochemistry
-
health nutrition
-
health kidney
-
What happens to body proteins when dietary energy is insufficient?
Body proteins are broken down into amino acids.
nutrition proteins -
nutrition energy
-
nutrition liver
-
What is the effect of using proteins for energy on the body's functional protein pool?
It reduces the body's functional protein pool, weakening muscle mass and enzyme activity.
nutrition muscle -
What happens to excess amino acids when protein intake is high?
Excess amino acids are deaminated (removal of the amino group).
nutrition proteins -
What can carbon skeletons from deaminated amino acids be oxidized for?
Carbon skeletons can be oxidized for ATP if energy demand exists.
nutrition energy -
What happens to carbon skeletons when energy and protein intake exceed needs?
They can be converted into fatty acids and stored as triglycerides.
nutrition fat -
Where are fatty acids stored when energy intake is high?
Fatty acids are stored as triglycerides in adipose tissue.
nutrition adipose_tissue -
What must be removed from an amino acid before it can be used for ATP production?
The nitrogen group
biochemistry metabolism -
biochemistry coenzymes
-
nutrition vitamins
-
nutrition deficiency
-
biochemistry atp
-
biochemistry atp
-
biochemistry atp
-
biochemistry metabolism
-
biochemistry coenzymes
-
nutrition vitamins
-
The amount of ATP formed by the catabolism of an amino acid carbon skeleton depends on where it entered the catabolism pathways.
biochemistry metabolism -
The carbon skeleton of an amino acid can be converted into pyruvate, acetyl-CoA, or intermediates of the citric acid cycle.
biochemistry metabolism -
biochemistry metabolism
-
What are amino acids that can be converted to pyruvate or an intermediate in the citric cycle called?
Glucogenic amino acids
biochemistry amino_acids -
biochemistry amino_acids
-
biochemistry amino_acids
-
biochemistry amino_acids
-
Amino acids that can be converted to pyruvate or an intermediate in the citric cycle are called glucogenic amino acids.
biochemistry amino_acids -
biochemistry amino_acids
-
biochemistry amino_acids
-
biochemistry amino_acids
-
The following amino acids are ketogenic: - Leucine - Lysine - Isoleucine - Phenylalanine - Tyrosine.
biochemistry amino_acids -
biochemistry metabolism
-
biochemistry metabolism
-
What are some intermediates in the citric cycle related to amino acid metabolism?
- Pyruvate
- Acetyl CoA
- Oxaloacetate
- Alpha-ketoglutarate
biochemistry citric_cycle -
biochemistry diagrams
-
nutrition biology
-
What is negative nitrogen balance?
The body loses more nitrogen than it retains; more protein is broken down than synthesized.
nutrition biology -
nutrition biology
-
What is positive nitrogen balance?
The body retains more nitrogen than it loses; more protein is synthesized than broken down.
nutrition biology -
When does positive nitrogen balance occur?
During rapid growth, recovery from illness, or weight training.
nutrition biology -
What is the relationship between nitrogen intake and output in positive nitrogen balance?
Nitrogen intake > nitrogen output; total body protein increases.
nutrition biology -
nutrition biology
-
nutrition biology
-
What is the effect of elevated protein intake over long periods of time?
- Kidney Stress
- Bone health issues
- Increased risk of heart disease and cancer
nutrition health -
nutrition diet
-
Elevated protein intake can lead to issues such as kidney stress, bone health issues, and an increased risk of heart disease and cancer.
nutrition health -
nutrition malnutrition
-
nutrition kwashiorkor
-
nutrition marasmus
-
nutrition pem
-
What is Kwashiorkor?
A disease typically affecting children caused by protein deficiency despite adequate energy intake.
nutrition disease -
What are the symptoms of Kwashiorkor?
- Stunted growth
- Poor immunity
- Bloated belly due to fat accumulation in the liver
symptoms health -
Why does fluid accumulate in the abdomen in Kwashiorkor?
Due to insufficient protein in the blood, which prevents water from diffusing out of blood vessels.
health physiology -
What is marasmus a result of?
A deficiency of energy; proteins and other nutrients are usually insufficient.
nutrition marasmus -
nutrition marasmus
-
nutrition kwashiorkor
-
nutrition eating_disorders
-
health allergy
-
health disease
-
What are common symptoms of a food allergy?
Hives, rashes, difficulty breathing, and potentially anaphylaxis.
health symptoms -
health treatment
-
What is food intolerance?
A non-immune mediated reaction, such as lactose intolerance due to lack of lactase.
health intolerance -
What is gluten sensitivity?
A condition with no known immune response, different from Celiac disease.
health sensitivity -
Do food intolerance and sensitivity involve an immune response?
No, they do not involve antibody production.
health immune -
What symptoms might indicate a food intolerance?
Bloating, diarrhea, and other digestive issues without an immune component.
health symptoms -
What are the functions of the immune system?
- Protects against infection
- Protects against cancer cells
- Promotes wound repair
- Recognizes 'foreign' agents as non-self
biology immune_system -
biology allergies
-
What are autoimmune diseases?
Diseases where the immune system aberrantly reacts to self, e.g., rheumatoid arthritis, multiple sclerosis.
biology autoimmunity -
biology immune_system
-
What does the innate immune system do?
Mediates initial protection against infections, always present, blocks entry of microbes, rapidly eliminates microbes that enter host tissues.
biology innate_immunity -
How does the adaptive immune system function?
Develops slowly, mediates later defense against infections, stimulated by invading microbes, adapts to their presence.
biology adaptive_immunity -
What is a food allergy?
An immune response to a food antigen, usually a protein, often involving IgE and the release of histamine and serotonin.
health food_allergies -
What typically causes a food allergy reaction?
A hypersensitivity to an antigen from a food source, generally a protein.
health food_allergies -
health food_allergies
-
health food_allergies
-
health food_allergies
-
Are food allergies inherited?
Yes, food allergies can be inherited, but not necessarily to a specific antigen.
health food_allergies -
What is the role of antigen-presenting cells in food allergies?
They recognize allergens and activate T-cells and B-cells.
immunology allergies -
immunology allergies
-
immunology allergies
-
What are common allergic symptoms caused by mast cells?
Symptoms include those caused by histamine and tryptase release.
immunology allergies -
The mechanism of food allergies involves the recognition of allergens by antigen-presenting cells, activation of T-cells and B-cells, and production of specific IgE.
immunology allergies -
immunology allergies
-
immunology allergies
-
What is the main function of B-cells in food allergies?
B-cells produce specific IgE in response to allergens.
immunology allergies -
What is the role of T-cells in the immune response to allergens?
T-cells are activated by antigen-presenting cells to help in the immune response.
immunology allergies -
What does the diagram illustrate about the immune response to allergens?
It shows allergen recognition, T-cell and B-cell activation, IgE production, and mast cell activation.
immunology allergies -
health allergies
-
health anatomy
-
health respiratory
-
health skin
-
What symptoms occur in the GI tract due to mast cell activation?
- Itching in mouth
- Trouble swallowing
- Nausea
- Vomiting
- Diarrhea
health gastrointestinal -
What is a life-threatening condition caused by mast cell activation in breathing & circulation?
Anaphylaxis
health emergency -
health emergency
-
health emergency
-
What are the Big 9 common food allergies?
- Shellfish
- Fish
- Peanuts
- Tree Nuts
- Eggs
- Wheat
- Soy
- Milk
- Sesame
food allergies -
food allergies statistics
-
What must be declared on food ingredients according to the US Food Allergens Act?
Common food allergens, specifically the Big 9.
food allergies regulations -
food allergies dates
-
food allergies antigens
-
What is the carbohydrate antigen related to red meat allergies?
Galili antigen (Galactose-alpha-1,3-galactose)
food allergies antigens -
health allergies
-
health allergies
-
health anaphylaxis
-
health anaphylaxis
-
health symptoms
-
health treatment
-
Peanut allergies are highly predominant in the US, affecting 1.8% of the population and often being severe and lifelong.
health allergies -
Anaphylaxis involves the immune system's release of chemicals that cause shock including symptoms like low blood pressure, airway narrowing, and blocked breathing.
health anaphylaxis -
health treatment
-
The diagram illustrates the process of sensitization and allergic reaction to an allergen, including immune cells and symptoms.
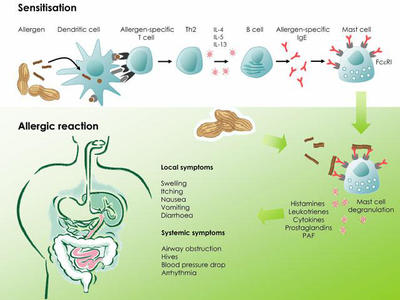 health illustration
health illustration -
What is celiac disease?
An autoimmune disorder where antibodies against gluten damage intestinal villi, leading to poor nutrient absorption.
health disease -
nutrition protein
-
nutrition protein
-
nutrition wheat
-
Celiac disease is an autoimmune disorder in which antibodies made against gluten react with intestinal villi, resulting in damage to the small intestine and poor absorption of nutrients.
health disease -
Gluten is a protein found in many grain-based foods that gives elasticity to dough and is composed of two main proteins: gliadin and glutenin.
nutrition protein -
nutrition wheat
-
What are common symptoms of celiac disease?
- Mild to severe abdominal pain
- Gas and bloating
- Chronic diarrhea
- Weight loss
- Poor growth (children)
health celiac_disease -
What serious conditions can result from untreated celiac disease?
- Anemia
- Osteoporosis
- Liver disease
- Intestinal cancer
- Dementia
health celiac_disease complications -
What tests are used to diagnose celiac disease?
- Blood tests
- Intestinal biopsies
- Genetic blood tests (if negative)
health diagnosis celiac_disease -
health celiac_disease testing
-
health celiac_disease genetics
-
health celiac_disease biopsy
-
Signs of celiac disease include: - abdominal pain - gas and bloating - chronic diarrhea - weight loss - poor growth (children)
health celiac_disease -
health celiac_disease diagnosis
-
Celiac disease can lead to complications such as anemia, osteoporosis, liver disease, and intestinal cancer.
health celiac_disease complications -
Blood tests for celiac disease include: - anti-tissue transglutaminase - endomysial (EMA) - deamidated gliadin peptide (DGP)
health celiac_disease testing -
What is the difference between gluten sensitivity and Celiac disease?
Gluten sensitivity does not cause damage to the small intestine like Celiac disease does.
health nutrition -
What is Non-Celiac Gluten Sensitivity (NCGS)?
It refers to gluten or wheat sensitivity without having Celiac disease.
health nutrition -
What are some gastrointestinal symptoms of gluten sensitivity?
- Bloating
- Abdominal pain
- Diarrhea
- Nausea
health symptoms -
What are some extraintestinal symptoms of gluten sensitivity?
- Headaches
- Fatigue
- Joint pain
- Brain fog
- Depression
- Skin rashes
health symptoms -
health visuals
-
What are some foods to avoid that contain gluten?
- Barley
- Rye
- Triticale
- Wheat
- Wheat-enriched flour
- Durum flour
- Graham flour
- Semolina flour
- Farina
- Wheat bran
- Wheat germ
- Cracked wheat
- Wheat protein
nutrition gluten foods_to_avoid -
What are some generally safe foods that are gluten-free?
- Arrowroot
- Buckwheat
- Cassava
- Corn
- Flax
- Millet
- Nuts
- Oats (small amounts)
- Quinoa
- Rice
- Sorghum
- Soy
- Tapioca
nutrition gluten safe_foods -
Is the gluten-free diet promoted for weight loss?
Yes, but claims are not supported by scientific evidence.
diet gluten-free weight_loss -
Do gluten-free foods have more or fewer calories than similar gluten-containing foods?
Gluten-free foods often contain more calories due to the need for multiple ingredients to replace gluten.
diet gluten-free calories -
Can a poorly planned gluten-free diet lead to weight gain?
Yes, it may lead to weight gain rather than weight loss.
diet gluten-free weight_gain -
What are food intolerances characterized by?
Unpleasant physical reactions following consumption of certain foods.
health nutrition -
How is a food intolerance different from a food allergy?
A food intolerance does not trigger the body's immune response.
health nutrition -
What are common causes of food intolerance?
- Lactose
- Monosodium glutamate (MSG)
- Caffeine
- Short-chain fermentable carbohydrates (FODMAPs)
health nutrition -
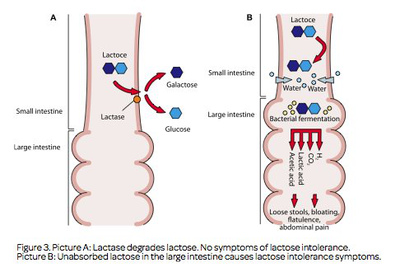
What is lactose intolerance?
The body cannot digest lactose; it is not an allergic reaction to milk.
health nutrition -
biology enzymes
-
biology anatomy
-
What happens to lactase production as we age?
The small intestine does not make enough lactase to break down lactose; lactase degrades over age.
biology aging -
What condition is referred to as lactase insufficiency?
A condition where the body does not produce enough lactase to digest lactose.
health nutrition -
health nutrition
-
biology enzymes
-
health nutrition animals
-
health digestion
-
health digestion
-
health digestion
-
health digestion
-
health digestion
-
health digestion
-
health digestion
-
health digestion
-
health digestion visuals
-
What happens to the lactase gene in lactose intolerant adults?
The gene that encodes lactase gets turned off.
biology genetics -
What percentage of the human population is truly lactose tolerant?
Only 35% of the human population is truly lactose tolerant.
health nutrition -
biology health
-
What does the Lactose Intolerance Treatment Market share data include for 2023?
It includes data by region: North America, Asia-Pacific, Europe, Middle East and Africa, South America.
market health -
What is the CAGR for the Lactose Intolerance Treatment Market projected from 2024-2030?
The market is projected to have a 7.1% CAGR from 2024-2030.
market growth -
What types of end-users are included in the Lactose Intolerance Treatment Market?
End-users include hospitals, homecare, specialty clinics, and others.
market health -
What type of infographic is provided about the Lactose Intolerance Treatment Market?
It shows market share by region, type, and end-user.
infographic market -
biology genetics
-
health nutrition
-
biology health
-
The Lactose Intolerance Treatment Market share data includes regions like North America, Asia-Pacific, Europe, Middle East and Africa, and South America.
market health -
market growth
-
End-users in the Lactose Intolerance Treatment Market include hospitals, homecare, specialty clinics, and others.
market health -
The infographic about the Lactose Intolerance Treatment Market shows market share by region, type, and end-user.
infographic market -
Lactase persistence means that the lactase protein continues to be produced in adults. This occurs due to an epigenetic modification in the lactase gene, which is typically switched off in late childhood.
biology genetics -
What is the significance of SNP ID: rs4988235 T/T?
It is often used as a predictor for dairy intake and is responsible for lactase persistence in people of European descent.
genetics lactase snp -
What happens to lactase production in late childhood?
The lactase gene is typically switched off, leading to decreased lactase protein production.
biology lactase -
What is lactase persistence?
It is the continued production of the lactase protein in adults, allowing them to digest lactose.
biology lactase -
biology enzymes
-
What is a common genetic variation in dairying populations?
A single nucleotide polymorphism (SNP) often associated with lactase persistence.
genetics dairy -
What is Phenylketonuria (PKU)?
A genetic metabolic disorder that prevents the conversion of phenylalanine into tyrosine, leading to phenylalanine accumulation.
genetics metabolism -
biochemistry pku
-
neuroscience pku
-
health newborns
-
biochemistry enzymes
-
nutrition pku
-
In normal metabolism, phenylalanine is converted to tyrosine. In PKU, this reaction is blocked, leading to the production of phenylketones that are toxic to the brain.
biochemistry metabolism -
Diagram (a) illustrates the metabolism of phenylalanine. In normal conditions, phenylalanine hydroxylase converts phenylalanine to tyrosine. In PKU, this conversion is blocked, leading to the accumulation of phenylalanine.
biochemistry pku -
In PKU, there is insufficient or malformed phenylalanine hydroxylase, which blocks the conversion of phenylalanine to tyrosine.
biochemistry pku -
What is the purpose of an elimination diet?
To identify foods causing allergic reactions or digestive issues by eliminating specific foods and gradually reintroducing them.
diet nutrition health -
Who should ideally guide an elimination diet?
A healthcare professional to ensure nutritional balance and safety.
diet healthcare nutrition -
What does an elimination diet allow individuals to do?
Create a customized diet that improves health and well-being.
diet customization health -
What is the first step in an elimination diet?
Eliminate specific foods from the diet for a period of time.
diet elimination steps -
What are some foods to eliminate in an elimination diet?
Dairy, gluten, soy, eggs, peanuts, shellfish, corn, tree nuts, fish, preservatives, artificial sugars, and artificial dyes.
diet foods elimination -
What is the goal of the Easy Elimination Diet?
To identify trigger foods through a phased approach of elimination and reintroduction.
diet easy_elimination trigger_foods -
The elimination diet is designed to identify foods that may cause allergic reactions, food intolerances, or other digestive issues.
diet nutrition health -
Individuals should ideally conduct an elimination diet under the guidance of a healthcare professional to ensure nutritional balance and safety.
diet healthcare nutrition -
The Easy Elimination Diet includes phases that detail specific food groups to eliminate and reintroduce over time.
diet easy_elimination phases -
Foods to eliminate in an elimination diet may include: dairy, gluten, soy, eggs, peanuts, shellfish.
diet foods elimination -
The goal of an elimination diet is to gradually reintroduce foods to determine which ones trigger symptoms.
diet symptoms trigger_foods -
The first step in an elimination diet is to eliminate specific foods from the diet for a period of time.
diet elimination steps -
schedule discussion food_allergies
-
reading chapter
-
resources canvas






 What animal is often associated with lactose intolerance?
What animal is often associated with lactose intolerance?