Sign up to unlock more features
- Save this deck to your account
- Study flashcards with spaced repetition
- Export to Anki (.apkg) or PDF
- Process documents up to 100 pages
- Images extracted from PDFs and documents
- Better text extraction from your PDFs and documents
- Better flashcards with our more advanced AI model
Flashcards in this deck (108)
-
What is the naming order for binary ionic compounds?
The cation is named first, followed by the anion.
chemistry nomenclature -
chemistry nomenclature
-
chemistry nomenclature
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
chemistry cations
-
chemistry anions
-
What do group 4A elements prefer in bonding?
They prefer to share electrons (covalent) than gain/lose 4.
chemistry bonding -
Should prefixes be used with the number of atoms in binary ionic compounds?
No, do not put prefixes; you can calculate the number needed for neutral charge.
chemistry nomenclature -
What is shown in Table 2.3?
It lists common monatomic cations and anions with their formulas and names.
chemistry tables -
chemistry ions
-
chemistry ions
-
chemistry compounds
-
chemistry compounds
-
chemistry ions
-
What are binary ionic compounds?
Compounds formed from two different elements, typically a metal and a non-metal.
chemistry compounds -
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry cations
-
What are common Type II cations?
Cations that can have multiple charges (e.g. Fe\(^{2+}\), Cu\(^{2+}\)).
chemistry cations -
chemistry anions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
What is the significance of Roman numerals in metal ion charges?
They specify the charge of metals that can form more than one type of positive ion.
chemistry ions -
What does the image represent?
A periodic table section highlighting common Type I cations, Type II cations, and monatomic anions.
chemistry visual -
What is the naming convention for the ion with higher charge?
Ends in -ic. Example: Fe3+ is ferric ion.
chemistry ions -
What is the naming convention for the ion with lower charge?
Ends in -ous. Example: Fe2+ is ferrous ion.
chemistry ions -
How do you determine the charge of a metal ion in compounds?
By balancing the positive and negative charges in the compound.
chemistry ions -
chemistry ionic_compounds
-
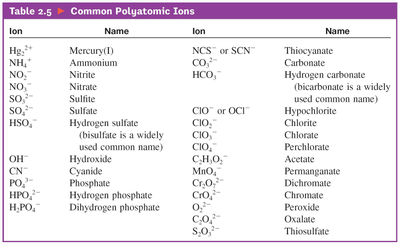
chemistry polyatomic_ions
-
chemistry polyatomic_ions
-
chemistry ionic_compounds
-
chemistry ionic_compounds
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry polyatomic_ions
-
chemistry visual_aid
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry ions
-
chemistry terms
-
chemistry nomenclature
-
chemistry nomenclature
-
chemistry nomenclature
-
chemistry nomenclature
-
chemistry terms
-
chemistry ions reference